Molecular Detection of Complex Vertebral Malformation in Holstein Calves
By
2014, Vol. 6 No. 05 | pg. 1/2 | »
IN THIS ARTICLE
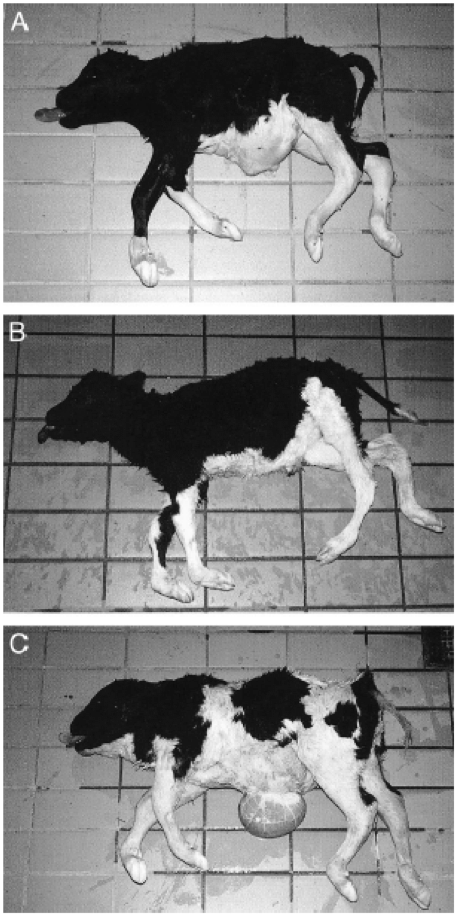
AbstractComplex vertebral malformation (CVM) in Holstein calves is a recessive syndrome that is the result of a transversion mutation within the SLC35A3 gene (Thomsen et al. 2006). Though a simple point mutation, this transversion results in high embryonic mortality and severe phenotypic abnormalities in surviving calves, such as growth retardation, vertebral malformation, and arthrogryposis (Agerholm et al. 2001). It is important to identify CVM carriers within the dairy market in order to effectively remove them from breeding centers and reduce the number of offspring with CVM. (Wang et al. 2012). CVM carriers possess increased milk production and decreased rates of conception when compared to non-carriers (Qin et al. 2010; Ghanem et al. 2008). Several molecular detection methods for CVM carriers have been developed since the discovery of the syndrome in Holstein calves, including created restriction site PCR, PCR-primer induced restriction analysis, mismatch amplification mutation assay-PCR, high resolution melting analysis, and PCR single stranded conformation polymorphism (Wang et al. 2011; Mahdipour et al. 2010; Gaber et al. 2012; Wang et al. 2012; Rusc and Kaminski 2007). Using these molecular approaches, research has effectively detected hundreds of CVM carriers within dairy markets and removed them from breeding, eradicating carriers within a matter of years (Rusc et al. 2013). It has become important to apply molecular methods to detect CVM carriers in dairy markets due to increased prevalence of such carriers, and perfecting those methods will be essential in ensuring accurate detection (Gabor et al. 2012). Complex vertebral malformation (CVM) in Holstein calves is a relatively recent discovery, with the syndrome’s initial characterization occurring in 1999 after Denmark breeders noticed a constant set of malformations in calves (Agerholm et al. 2001). However, it was not until 2001 that researchers were able to publish results on CVM in a group of Holstein calves (Agerholm et al. 2001). Much of the early research conducted on CVM in Holstein calves focused on the phenotypic variation as well as on the basic genetic aspects of the condition. It was only recently that more detailed genetic analyses were conducted to further assess the syndrome. CVM is a homozygous recessive syndrome caused by a mutation at position 559 of the solute carrier family 35 member 3 gene, SLC35A3, which is located on bovine chromosome BTA3 (Thomsen et al. 2006). The phenotypic variation among Holstein calves with CVM is significant, though there are some unifying characteristics of the syndrome that have allowed for relatively easy visual detection. Among most cattle, growth retardation is common, in addition to varying degrees of vertebral malformation (such as vertebral lesions), arthrogryposis of several joints (which limits the mobility of the cattle), and cardiac abnormalities involving the septum and aorta. As shown in Figure 1, calves born with CVM can exhibit short vertebral columns and scoliosis (Figure 1a and 1b), while some even have herniation of certain stomach compartments (Figure 1c) (Agerholm et al. 2001). The mortality of affected cattle is significant. Nielsen et al. (2002) assessed the survival rate of fetuses homozygous for CVM by crossing female Hostein cows who were carriers for the syndrome with males who were either normal or carriers. Based on data from 62 inseminations, they observed that 77% of the fetuses that were identified to be homozygous for CVM were aborted before the normal gestational age of 260 days (Nielsen et al. 2002).CVM provides an example of the risks of inbreeding cattle despite the benefits to the dairy market.
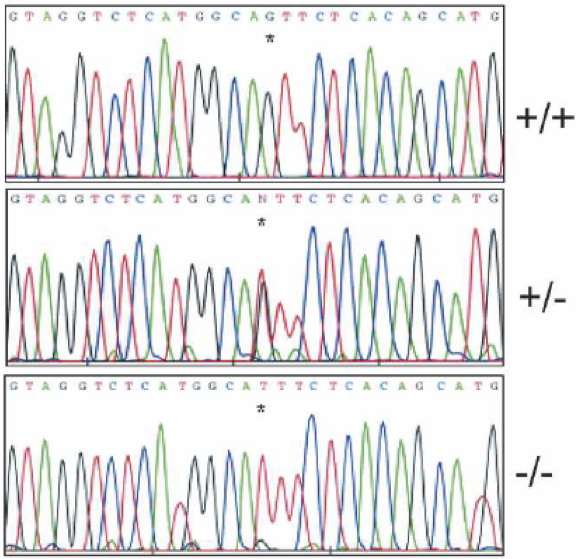
Figure 1: Phenotypic variations in Holstein calves with CVM. Growth retardation and vertebral malformation are common in calves born with CVM. A and B are calves that exhibited short vertebral columns and scoliosis, while C is a calf that exhibited herniation of stomach compartments (From Agerholm et al. 2001). Simple genetic approaches were used initially to identify the mutation responsible for the development of CVM in Holstein calves. Thomsen et al. (2006) compared cDNA sequences between SLC35A3 wild type and mutated cattle, identifying that the mutated cattle possessed a transversion from a guanine to a thymine within the SLC35A3 gene that did not affect the splicing of the resulting mRNA strands (Figure 2) (Thomsen et al. 2006). Though a small mutation, a resulting conversion of valine to phenylalanine in the protein product, Uridine diphosphate N-acetylglucosamine transporter (UDP-GlcNAc), occurred in the affected Holstein calves. UDP-G1cNAc is a nucleotide sugar that is transported across the Golgi membrane in cattle cells by the protein product transcribed from SLC35A3, a function which is critical for the maintenance of cytoskeleton structure. (Patel 2012). Thomsen et al. (2006) also conducted an assay in yeast culture to identify the potential effect of the mutation on SLC35A3 gene expression in comparison to the wild-type gene, but Northern blotting revealed no significant differences in expression levels. Consequently, there is still no definitive answer as to how the mutation of SLC35A3 influences the transport of UDP-G1cNAc into the Golgi membrane, although it is apparent that the impact of the mutation is disastrous due to the multiple and varying phenotypic abnormalities that result from this single mutation to the Holstein calf genome (Thomsen et al. 2006). Perhaps most critical in the early genetic analyses of CVM was the difference between heterozygous carriers and homozygous calves. While heterozygous carriers for the SLC35A3 gene are unaffected by the syndrome and were thought to develop normally, the small proportion of surviving homozygous calves are so severely malformed that they are typically euthanized (Agerholm et al. 2004). However, the presence of a small proportion of cattle who do survive beyond birth, even if malformed, led Thomsen et al. (2006) to postulate the homozygous genotype is not entirely lethal. Specifically, the mutation to SLC35A3 still might produce a partially functional UDP-G1cNAc transporter or induces the use of a similar transporter (Thomsen et al. 2006).
Figure 2: Nucleotide sequences for SLC35A3 gene in wild type, carrier, and affected Holstein bulls; +/+ represents non-carriers, +/- represents carriers, and -/- represents cattle with CVM (From Thomsen et al. 2006). RelevancePrior to the development of CVM, it was common for large-scale inbreeding to occur among cattle in order to promote the transmission of superior genes to offspring who would provide a source of dairy (Patel 2012). Patel (2012) describes the history of the syndrome, finding that a single ancestral bull that was heterozygous for CVM was responsible for the widespread proliferation of the syndrome through inbreeding. CVM provides an example of the risks of inbreeding cattle despite the benefits to the dairy market. CVM in Holstein calves has significant repercussions for milk production in carriers, which do not show any obvious physiological deformities. In a study that compared carriers and non-carriers, Qin et al. (2010) identified that carriers for CVM were better milk producers than were non-carriers based on several milk production traits, with statistically significant results based on hundreds of Holstein calves studied. The implication for this research is the desirability of carriers for CVM in the dairy market, but of course selective breeding is critical so that homozygous calves with the severe malformations of CVM are not produced. Thus, detection techniques for carriers for the purpose of selective breeding are necessary if the dairy market chooses to raise CVM carriers, or to eradicate the mutated CVM gene to prevent any offspring with CVM (Wang et al. 2012).Continued on Next Page » Suggested Reading from Inquiries Journal
Inquiries Journal provides undergraduate and graduate students around the world a platform for the wide dissemination of academic work over a range of core disciplines. Representing the work of students from hundreds of institutions around the globe, Inquiries Journal's large database of academic articles is completely free. Learn more | Blog | Submit Latest in Biology |