In SummaryGene Therapy: Current Treatment Options and Likely Near-Term Developments
By
2014, Vol. 6 No. 09 | pg. 1/2 | »
IN THIS ARTICLE
KEYWORDS
AbstractGene therapy has ceased to be a futuristic dream and instead become a treatment for the present. Somatic cells can be treated with gene replacements or gene fragments to express required proteins and whilst gene therapy was originally aimed at inherited disease, it is now capable of treating a number of acquired problems such as cancer and HIV. Originally delivered through viruses, there are now safer direct or biochemical vectors while in the future nano-particles might deliver the drug of choice straight to the nucleus of affected cells. Gene therapy is used to correct inherited disorders, treat cancers and introduce genetic material to provoke an immune response (Ritter, Lehmann, & Volk, 2002), all of which are difficult to achieve with conventional treatments (Büning, 2013; Cross & Burmester, 2006; Scherer & Rossi, 2011). Since the 1990s, work has been carried out to improve gene therapy as a treatment model (Culver & Blaese, 1994) and the European Commission granted its first license for a gene therapy medicine in 2012 (Büning, 2013). 
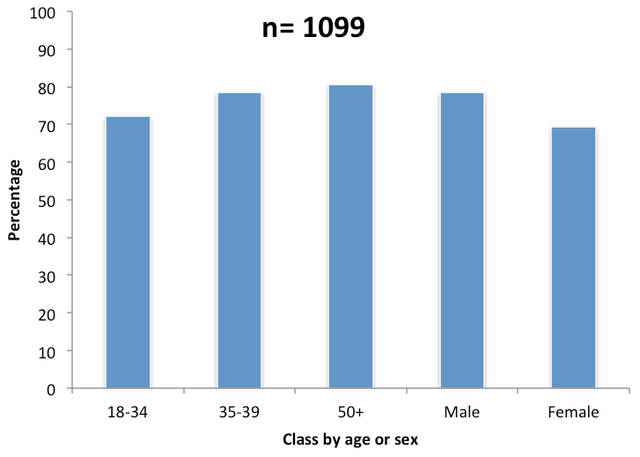
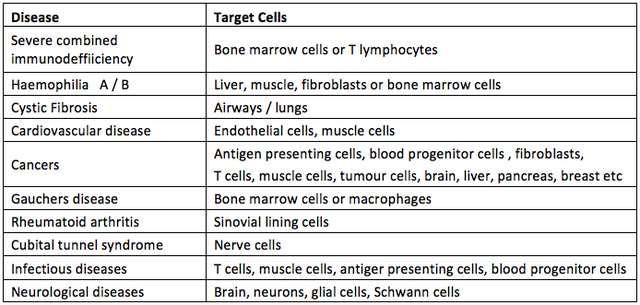
This article briefly discusses some of the materials used in gene therapy and their delivery methods. It reviews the types of disorders for which gene therapy may be useful and compares the advantages and disadvantages of various delivery systems for gene therapy. What Can Be Treated?Gene therapy cells usually treats somatic cells and although originally devised to treat inherited disorders (Evans & Robbins, 1995), most gene therapy now benefits cancer patients. It is also used to treat acquired immune disorders, arthritis and hemophilia (Evans & Robbins, 1995; Mountain, 2000). Gene therapy is an attractive proposition since certain Ribonucleic acids (RNAs) or proteins provide effective treatments but are traditionally difficult to deliver effectively. Conventional treatments for some disorders are invasive, unpleasant, frequently ineffective and often have side effects; gene therapy may allow for treatments without these difficulties. In the case of human acquired immunodeficiency virus (HIV), outcomes have been improved with the use of anti-retroviral drugs (Marathe & Wooley, 2007) but side effects can be unpleasant while current treatments represent disease management rather than any type of cure (Scherer & Rossi, 2011). The virus also mutates swiftly (Campbell & Reece, 2008) and many patients infected with HIV are found to have strains which have developed immunity to one or more of the available drugs (Hirsch et al., 2008; Richman et al., 2004) (Fig 1). Given that in 2011 the UK had an estimated 96,000 people living with HIV (Anon, 2013), the search for a cure is as urgent as ever and it is hoped that gene therapy may provide this cure (Marathe & Wooley, 2007). Treatment SitesSome treatments can be carried out ex vivo and others are only successful in vivo. Ex vivo therapy has been trialled for HIV (Scherer & Rossi, 2011) and Alzheimer’s disease (Tuszynski, Thal, Pay, & Salmon, 2005) amongst others and in vivo therapy has been trialled on Cystic Fibrosis and the majority (but not all) of cancer trials have been in vivo (Mountain, 2000). Delivery may be systemic or local, in local delivery the gene products are delivered directly to the site of treatment, for example to the synovial tissues in the treatment of rheumatoid arthritis (Evans and Robbins, 1995) whereas systemic delivery involves the treatment being applied to more than one site via the circulation; this system has been used to treat lung cancer (Gopalan et al., 2004). Treatment needs to be targeted at different cell types depending on the disorder to be treated, some of which are shown at Table 1. Once they are delivered the gene products may replace a faulty gene (for a genetic disorder) (Verma & Somia, 1997) or they may be used to illicit an immune response to enable killer cells to deal with a tumor (Cross & Burmester, 2006). Gene therapy can even deliver products to code for the production of proteins involved in healing broken bones (Evans & Robbins, 1995); the list is seemingly endless. Delivery MethodsThe biggest challenge facing clinicians in the development of gene therapy is the gene delivery method (Somia & Verma, 2000). Getting the genetic material to the site of treatment is only part of the problem, it then needs to be taken up and expressed (Evans& Robbins, 1995); this is achieved using vectors. In order to ensure delivery of the gene of interest vectors must be accurate and efficient (Miller & Vile, 1995), there is little point in delivering genetic therapies to the wrong cells in the patient. Vectors can be broadly divided into two groups; viral and non-viral (Evans & Robbins, 1995).
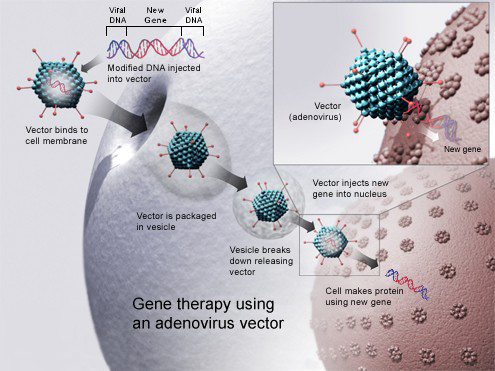
Viral SystemsViruses are evolved to and efficient at, getting their DNA into host cells (Thomas, Ehrhardt, & Kay, 2003) and so are potentially powerful tools in the vectoring of gene products into cells (Verma & Somia, 1997). The problem with this is that viruses are normally pathogenic and so they are usually disabled in some way before they are packaged with the gene(s) of interest to be vectored into the patient (Verma & Somia, 1997). This is done by removing the coding regions of the viral genome which allow for replication and host damage and leaving in place the sequences needed to repack the virus with the gene of interest and those required for integration and expression in the host DNA (Thomas et al., 2003). The viral vector is then delivered by infecting the host cell (Verma & Somia, 1997) . Normally the expression of genes is reliant upon the DNA getting access to the cell nucleus where the transcription apparatus resides. Expression can occur after the DNA is integrated into the chromosomal DNA of the host cell, in which case effects are long lasting and passed to daughter cells after division or it can occur without the DNA being absorbed (so called episomal DNA) in which case initial expression is often high but tails off, especially after cell division (Evans & Robbins, 1995). There are several classes of virus used as vectors and they fit into two main groups depending on whether they integrate DNA into the host chromatin or if their DNA remains extra-chromosomal within the host nucleus (Table 2) (Thomas et al., 2003). Figure 1. Percentage of US HIV sufferers presenting with virus immunity to at least one drug treatment (Richman et al., 2004). Table 1. Some of the diseases suitable for treatment with gene therapy and the target cells/sites involved (data from (Verma and Somia, 1997; Mountain, 2000). Each of these vectors has advantages and disadvantages (Table 2) and there are a number of other viruses including simian, papilloma and vaccinia which have potential in gene therapy but aren’t discussed here. Each virus will be suited to different applications; however discussion of those applications is beyond the scope of this review. The pathogenic origins of viral vectors have raised safety fears (Kay, Glorioso, & Naldini, 2001; Mountain, 2000; Thomas et al., 2003; Verma & Somia, 1997). Several approaches have been taken to reduce the dangers including the use of viruses from non-human sources (such as bovine lentiviruses) and self-disabling retroviruses (Kay et al., 2001) but dangers remain and chief amongst these is the possibility of mutagenesis leading to the switching on of an oncogene or deletion of a tumor suppressing gene either of which could result in a malignant tumor (Evans & Robbins, 1995). In 1999 an adenovirus vector caused the death of a patient and in 2002/3 two children treated with a retrovirus vector developed cancer (Thomas et al., 2003).Continued on Next Page » Suggested Reading from Inquiries Journal
Inquiries Journal provides undergraduate and graduate students around the world a platform for the wide dissemination of academic work over a range of core disciplines. Representing the work of students from hundreds of institutions around the globe, Inquiries Journal's large database of academic articles is completely free. Learn more | Blog | Submit Latest in Biology |