A Study of Planarian Phototaxis, Tactile Response and Regeneration
By
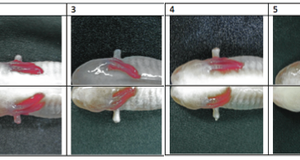
2014, Vol. 6 No. 03 | pg. 2/2 | « DiscussionThe “parent” Dugesia flatworm obtained from the stock culture successfully regenerated into two complete planarians with eyespots, auricles, and developed heads and tails after being decapitated. This conclusion is based on close observation of both planarians after 7 days of incubation. In addition to regenerating when decapitated, the “parent” planarian exhibited behaviors typical to planarians as indicated in the literature; it demonstrated a negative phototaxis response as well as tactile sensitivity (moving away from the source of touch, in the case of this experiment a pipette tip). Though it was not possible to assess any genetic or molecular mechanisms associated with the planarian regeneration (as much of the literature contains many of such studies), a good body of observable information was obtained during the course of the experiment. Furthermore, there are several important points to note based on the observations of this experiment. Firstly, although the planarian seems to have successfully regenerated into two full planarians, neither of the newly regenerated planarians reached the stretched length of 14 millimeters of the “parent.” This may be due to the fact that not enough time passed for both of the planarians to completely regenerate, even though they looked as if they did from a visual examination. Previous research indicates that it can take at least five days for planarian nervous system regeneration to occur (Inoue et al., 2004), so it is possible that the regenerated planarians were not fully developed by the time the final measurements were taken. Phototactic and tactile response behaviors should have also been observed after 7 days to assess full nervous system regeneration. Furthermore, the experimental findings indicate that regeneration in planarians requires time. This experiment showed that only after 7 days did the planarians seem to have complete regeneration (with the possibility of even more growth possible, based on the previous discussion of the length observations after 7 days). The time required for regeneration may be due to the various literature citations about genes and proteins critical for regeneration; it is the up-regulation of these genes and action of these proteins that acts as a mechanism to regenerate the decapitated planarian, and it is possible that this mechanism takes time to act within the planarian body. This suggests that planarians may need to exhibit some level of dormancy for the several days that it takes to completely regenerate.Additionally, a small amount of regeneration rate information can be derived from the minimal amount of measurements that were taken. Based on the linear regressions derived from Figure 1, the rate of regeneration appears to be about double that for the head region in comparison to the tail region (although measurements at equivalent increments of time were not taken) . However, there are not a significant number of measurements that were taken, nor were the differences in growth significant enough after 30 minutes of decapitation between the head and tail regions (approximately 1mm), so it is not possible to make any substantial conclusions regarding rates of regeneration of the head and tail regions. There is also not any significant information available in the literature about growth rates, so future studies should focus on this interesting aspect of planarian regeneration in order to contribute to the body of knowledge already available about planarians. In conclusion, the Dugesia planarian was successfully regenerated into two distinct planarians after decapitation. This study of the regenerative system of planarians is important because such studies provide insight into regenerative capabilities of other organisms as well. Although more length measurements and a final observation of potential phototactic and tactile responses after 7 days of regeneration would have been ideal, the experiment still shed light on the various characteristics of the planarian. Future studies should focus on growth rates of planarians as the undergo regeneration in order to fully comprehend the mechanisms involved in this complex but interesting process. ReferencesCebria, F. 2007. Regenerating the central nervous system: how easy for planarians! Developmental Genes and Evolution, 217: 733-748. Farnesi, R.M. & Tei, S. 1985. The frontal organ of a triclad flatworm, Dugesia lugubris. Cell and Tissue Research, 240-129-135. Gurley, K.A., Rink, J.C., & Alvarado, A.S. 2008. β-Catenin Defines Head Versus Tail Identity During Planarian Regeneration and Homeostasis. Science, 319: 323-327. Inoue, T., Kimamoto, H., Okamoto, K., Umesono, Y., Sakai, M., Alvarado, A.S., & Agata, K. 2004. Morphological and Functional Recovery of the Planarian Photosensing System during Head Regeneration. Zoological Science, 21: 275-283. Newmark, P.A. & Alvarado, A.S. 2000. Bromodeoxyuridine Speciï¬�cally Labels the Regenerative Stem Cells of Planarians. Developmental Biology, 220: 142-153 Reddien, P.W., Oviedo, N.J., Jennings, J.R., Jenkin, J.C., & Alvarado, A.S. 2005. SMEDWI-2 Is a PIWI-Like Protein That Regulates Planarian Stem Cells. Science, 310-1327-1330. Ruppert, E. E., Fox, R. S., &Barnes, R.D. 2004 Invertebrate Zoology: A functional evolutionary approach. Thomson Learning Takano, T., Pulvers, J.N., Inoue, T., Tarui, H., Sakamoto, H., Agata, K., & Umesono, Y. 2007. Regeneration-dependent conditional gene knockdown (Readyknock) in planarian: Demonstration of requirement forDjsnap-25expression in the brain for negative phototactic behavior. Development, Growth, and Differentiation, 49: 383-394. Umesono, Y. & Agata, K. 2009. Evolution and regeneration of the planarian central nervous system. Development, Growth, and Differentiation, 51: 185-195. Suggested Reading from Inquiries Journal
Inquiries Journal provides undergraduate and graduate students around the world a platform for the wide dissemination of academic work over a range of core disciplines. Representing the work of students from hundreds of institutions around the globe, Inquiries Journal's large database of academic articles is completely free. Learn more | Blog | Submit Latest in Biology |