In SummaryGene Therapy: Current Treatment Options and Likely Near-Term Developments
By
2014, Vol. 6 No. 09 | pg. 2/2 | « Non-Viral SystemsIt is against the backdrop of these safety concerns that non-viral vectors have been developed. Non-viral methods have several potential advantages over viral methods including risk free repeat administration (Lam & Dean, 2010), relative ease of large scale production and low host immunogenicity (Al-Dosari & Gao, 2009). Historically low levels of transfection and expression, caused by inability to breach cellular barriers and access the host DNA, have held non-viral vectors back (Atkinson & Chalmers, 2010) and as Lam and Dean (2010) point out; viral vectors have had millennia to evolve efficient methods of infecting hosts with their DNA, non-viral methods have not. The issue is that the gene of interest must get past the membrane of the nucleus and into the host chromatin without being damaged, in order to be expressed; this is now achievable in a number of ways. Table 2. Some of the major types of viral vector used in gene therapy with DNA incorporation type and summary of main advantages and disadvantages. Data from (Mountain, 2000; Kay et al., 2001; Thomas et al., 2003). 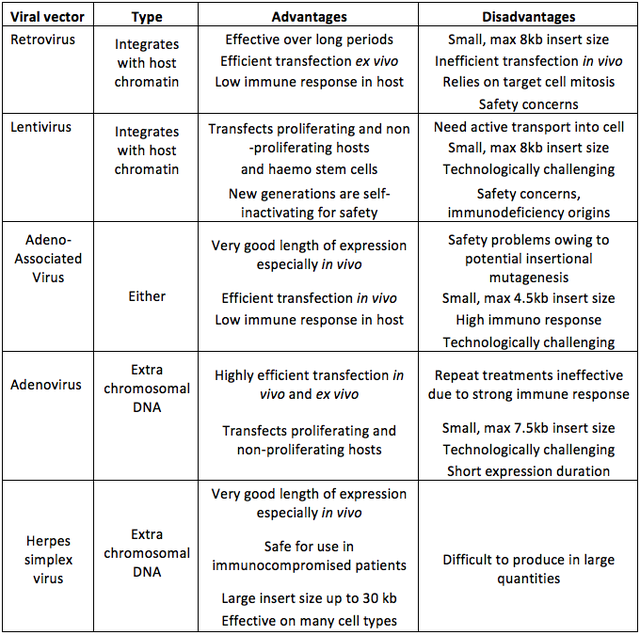
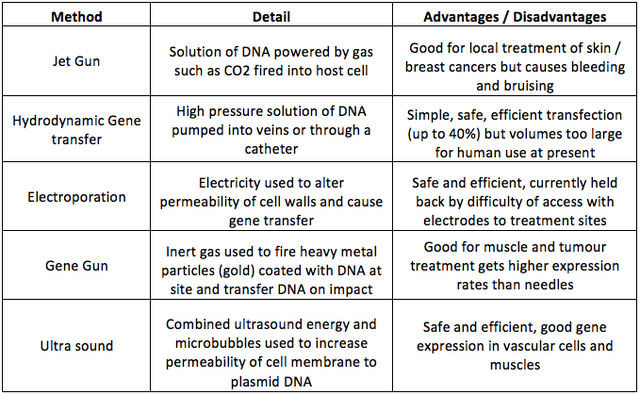
The simplest method of non-viral transfection uses naked DNA and this was first demonstrated in vivo when Wolff et al. (1990) injected protein expression vectors directly into murine models, although the effects of this were fairly long lasting they were only very local. There are a number of other non-viral methods of delivering naked DNA each of which has advantages and disadvantages (Table 3). Another promising non-viral route involves biochemical vectors. Like other non-viral routes these have low toxicity and low immunogenicity (Al-Dosari & Gao, 2009). The plasmid DNA is packaged in cationic lipids (often cholesterol) which bond well with anionic DNA and also interact with the cell membrane allowing the whole complex access to the cytoplasm where the lipids are absorbed and the plasmid released (Niidome & Huang, 2002). A similar system exists using biocompatible, biodegradable polymers these can be administered intravenously or into airways by aerosol and are used to treat cancer and cystic fibrosis (Al-Dosari & Gao, 2009). Table 3. Summary of most common direct methods of integrating naked DNA directly into host cells. Data from (Niidome and Huang, 2002; Al-Dosari and Gao, 2009; Lam and Dean, 2010). The Future of Gene TherapyThe future must involve fewer viral vectors and increased use of direct insertion of naked DNA, or where that is not feasible, the use of biochemical vectors. Nano particles also offer potential as future vectors. Transfection rates will increase as specificity and cell targeting improves. However mutation of diseases, such as cancer and HIV, is likely to mean that the arms race between clinicians and diseases will continue unabated. AcknowledgementsThanks to Enrico Ferrari for guidance and comments on an earlier version of this work. ReferencesAlberts, B. (2010). Essential cell biology (Vol. 3, p. 23,28,60,731). New York London: Garland Science. Al-Dosari, M. S., & Gao, X. (2009). Nonviral gene delivery: principle, limitations, and recent progress. The AAPS Journal, 11(4), 671–681. Anon. (2013). HIV facts > Latest UK Statistics. Atkinson, H., & Chalmers, R. (2010). Delivering the goods: viral and non-viral gene therapy systems and the inherent limits on cargo DNA and internal sequences. Genetica, 138(5), 485–498. Büning, H. (2013). Gene therapy enters the pharma market: The short story of a long journey. EMBO Molecular Medicine, 5(1), 1–3. Campbell, N. A., & Reece, J. B. (2008). Biology. The Benjamin/Cummings series in the life sciences (Vol. 8th, pp. 1267, various). San Francisco London: Pearson. Cross, D., & Burmester, J. K. (2006). Gene therapy for cancer treatment: past, present and future. Clinical Medicine & Research, 4(3), 218–227. Culver, K. W., & Blaese, M. R. (1994). Gene therapy for cancer. Trends in Genetics, 10(5), 174–178. Evans, C. H., & Robbins, P. D. (1995). Current concepts review: possible orthopaedic applications of gene therapy. Journal of Bone and Joint Surgery-A-American Volumes, 77(7), 1103–1115. Gopalan, B., Ito, I., Branch, C. D., Stephens, C., Roth, J. A., & Ramesh, R. (2004). Nanoparticle based systemic gene therapy for lung cancer: molecular mechanisms and strategies to suppress nanoparticle-mediated inflammatory response. Technology in Cancer Research & Treatment, 3(6), 647. Hirsch, M. S., Günthard, H. F., Schapiro, J. M., Vézinet, F. B., Clotet, B., Hammer, S. M., … Pillay, D. (2008). Antiretroviral drug resistance testing in adult HIV-1 infection: 2008 recommendations of an International AIDS Society-USA panel. Clinical Infectious Diseases, 47(2), 266–285. Kay, M. A., Glorioso, J. C., & Naldini, L. (2001). Viral vectors for gene therapy: the art of turning infectious agents into vehicles of therapeutics. Nature Medicine, 7(1), 33–40. Lam, a P., & Dean, D. a. (2010). Progress and prospects: nuclear import of nonviral vectors. Gene Therapy, 17(4), 439–47. doi:10.1038/gt.2010.31 Marathe, J., & Wooley, D. (2007). Is gene therapy a good therapeutic approach for HIV-positive patients? Genetic Vaccines and Therapy, 5(1), 5. Miller, N., & Vile, R. (1995). Targeted vectors for gene therapy. The FASEB Journal, 9(2), 190–199. Mountain, A. (2000). Gene therapy: the first decade. Trends in Biotechnology, 18(3), 119–128. Niidome, T., & Huang, L. (2002). Gene therapy progress and prospects: nonviral vectors. Gene Therapy, 9(24), 1647–1652. Richman, D. D., Morton, S. C., Wrin, T., Hellmann, N., Berry, S., Shapiro, M. F., & Bozzette, S. A. (2004). The prevalence of antiretroviral drug resistance in the United States. Aids, 18(10), 1393. Ritter, T., Lehmann, M., & Volk, H.-D. (2002). Improvements in Gene Therapy. BioDrugs, 16(1), 3–10. Roitt, I. M., Delves, P. J., Martin, S. J., & Burton, D. R. (2006). Roitt’s essential immunology (Vol. 11th, p. 474). Oxford: Blackwell Science. Scherer, L. J., & Rossi, J. J. (2011). Ex vivo gene therapy for HIV-1 treatment. Human Molecular Genetics, 20(R1), R100–R107. Seeley, R. R., Stephents., T. D., & Tate, P. (2000). Anatomy & physiology. Student study art notebook (Vol. 5th). Boston, Mass.: WCB/McGraw Hill. Somia, N., & Verma, I. M. (2000). Gene therapy: trials and tribulations. Nature Reviews Genetics, 1(2), 91–99. Thomas, C. E., Ehrhardt, A., & Kay, M. A. (2003). Progress and problems with the use of viral vectors for gene therapy. Nature Reviews Genetics, 4(5), 346–358. Tuszynski, M. H., Thal, L., Pay, M., & Salmon, D. P. (2005). A phase 1 clinical trial of nerve growth factor gene therapy for Alzheimer disease. Nature Medicine, 11(5), 551–555. Verma, I. M., & Somia, N. (1997). Gene therapy-promises, problems and prospects. Nature, 389(6648), 239–242. Wolff, J. A., Malone, R. W., Williams, P., Chong, W., Acsadi, G., Jani, A. and, & Felgner, P. L. (1990). Direct gene transfer into mouse muscle in vivo. Science, 247(4949), 1465–1468. Suggested Reading from Inquiries Journal
Inquiries Journal provides undergraduate and graduate students around the world a platform for the wide dissemination of academic work over a range of core disciplines. Representing the work of students from hundreds of institutions around the globe, Inquiries Journal's large database of academic articles is completely free. Learn more | Blog | Submit Latest in Biology |