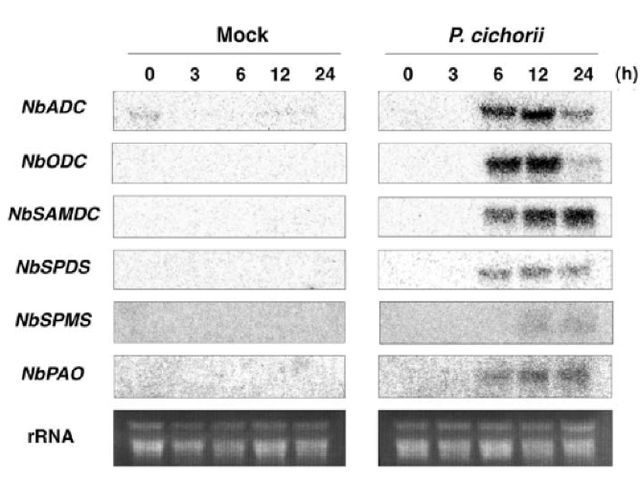
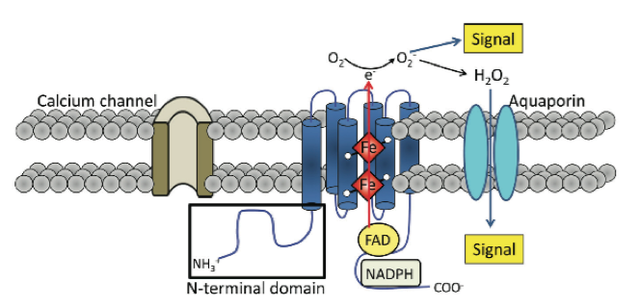
The Role of Hydrogen Peroxide in Controlling Plant Cell-Signaling and Gene-Expression Patterns Related to Stress and Defense ResponsesCURRENT WORKCurrent research into the role of hydrogen peroxide in plant stress and defense responses is primarily focused on uncovering novel sources of the compound in response pathways, examining molecular networks that control oxidative burst events, assessing interactions between various environmental and endogenous factors and H2O2 signaling, and studying gene products that protect cellular components during H2O2 responses. Efforts to uncover new sources of H2O2 have focused on one specific defense system known as the hypersensitive response (HR). During a HR, a necrotic lesion is formed at the location where the pathogen enters the plant body, serving as a blockade to the spread of the disease (Yoda et al. 2009). Yoda et al. (2009) demonstrated how tobacco plants infected with pathogenic Pseudomonas cichorii showed an accumulation of polyamines in apoplasts, and induction of transcripts for six genes coding for enzymes functioning in polyamine metabolism: NbPAO, NbODC, NbSAMDC, NbSPDS, NbSPMS, and NbPAO (Figure 4). Induction was confirmed in the infected group by creating an accumulation profile through RNA gel-blot analysis, which revealed greater accumulation of all six enzymes relative to a group of tobacco plants that received mock (water) treatment. Of the genes encoding the enzymes, the polyamine oxidase gene (NbPAO) was shown to be essential for hydrogen peroxide generation and subsequent HR. This was indicated by silencing of NbPAO through the virus-induced gene silencing method (VIGS), confirmed by real-time polymerase chain reaction, which resulted in low levels of hydrogen peroxide production, low HR, and increased bacterial growth upon pathogen challenge (Yoda et al. 2009). Sagor et al. (2012) revealed that heat shock in Arabidopsis caused the induction of a spermine synthase gene (SPMS), S-adenosylmethionine gene, and an arginine decarboxylase 2 gene. The corresponding gene products, including the common polyamine spermine, were shown to increase linearly with heat shock. The same study revealed that two extreme types of transgenic plants, one overexpressing SPMS and one SPMS knockout mutant (spms), showed upregulation of four genes coding for heat-shock proteins: HSP101, HSP90, HSP70, and HSP17.6. However, the plant with the greater spermine content showed greater expression of all four types, and consequently greater relative thermoresistance. Drerup et al. 2013 has also suggested that the genes modulating the oxidative burst might be part of a larger gene network, rather than independent entities within plant genomes. This multigenic family, referred to as Rboh, includes a variety of respiratory burst oxidase homologs, including the well-studied NADPH oxidases. The genes in the Arabidopsis Rboh family encode for integral membrane proteins characterized by six membrane-spanning domains, two heme groups and a C-terminal domain with NADPH- and FAD- binding sites (Figure 5). Furthermore, plant Rboh-encoded proteins also have extended N-terminal regions that contain calcium binding EF-hand motifs and phosphorylation domains, relevant to oxidase function (Drerup et al. 2013). Figure 4 – Accumulation profile of transcripts for 6 enzymes (NbPAO, NbODC, NbSAMDC, NbSPDS, NbSPMS, and NbPAO) responsible for polyamine metabolism during a hypersensitive response.
Wild-type tobacco leaves received either mock (water) treatment or P. cichorri for 0 hours, 3 hours, 6 hours, 12 hours, and 24 hours. RNA transcripts were extracted from each of the leaves in the experiment at each time interval, and a RNA gel-blot analysis was conducted with Phosphorus-32 labeled probe. rRNA served as the loading control for the gel-blot analysis. (From Yoda et al. 2009) Figure 5 – Typical structural organization of Arabidopsis respiratory burst oxidase homolog.
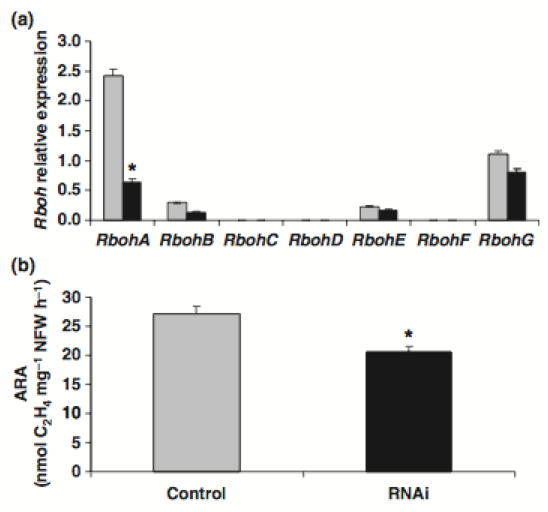
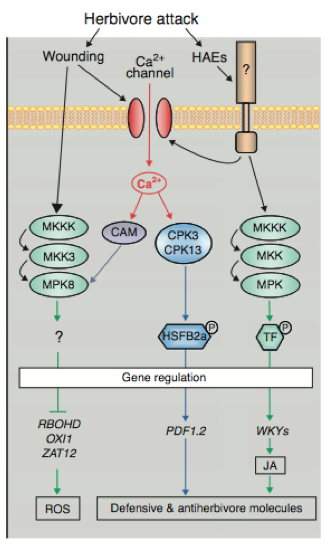
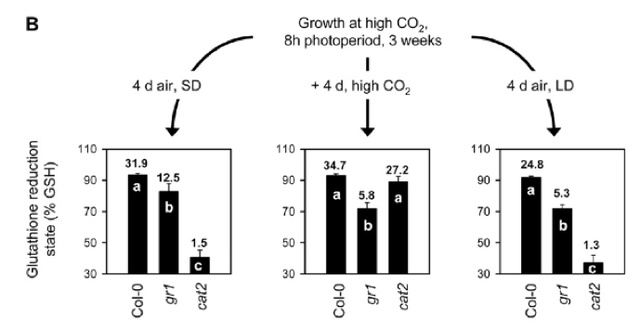
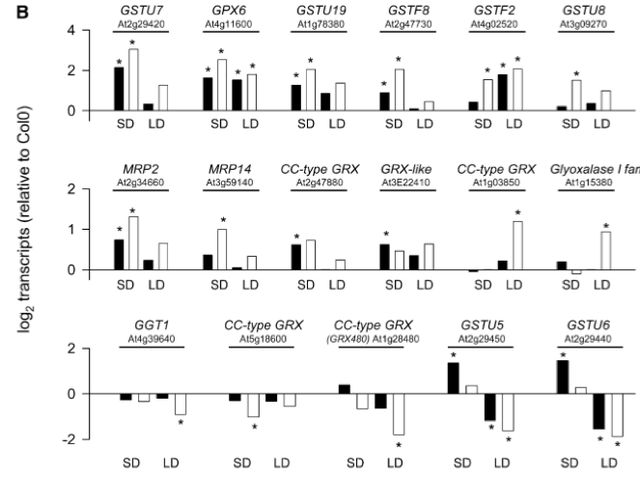
Structural representation of Arabidopsis Rboh family, which encodes for integral membrane proteins that characteristically have six membrane-spanning domains, two heme groups and a C-terminal domain with NADPH- and FAD- binding sites. (From Baxter et al. 2014) A study by Marino et al. (2011) with the legume Medicago truncatula further indicated the importance of these genes in the context of forming a symbiotic relationship with the bacterium Sinorhizobium meliloti. Using sequence similarity searches with previously identified Arabidopsis Rboh genes, Marino et al. (2011) identified seven putative RBOH-encoding genes in the Medicago genome with some duplications: MtRbohA, MtRbohB, MtRbohC, MtRbohD, MtRbohE, MtRbohF, and MtRbohG. Of these seven, MtRbohA was significantly up-regulated in symbiotic nodules that were induced by the bacterial species, an effect that was enhanced under hyperoxic conditions. As further confirmation of its role in establishing symbiotic connections, RNAi of MtRbohA significantly hindered symbiotic nodule formation by decreasing required nitrogen fixation activity of the bacterium, as measured by an acetylene reduction assay that measured the activity level of the enzyme nitrogenase in reducing acetylene gas to ethylene (Figure 6). Hydrogen peroxide presence and activity was also noted in the legumes, though the relationship between the two species was not antagonistic (Marino et al. 2011). This points to the possibility that H2O2 signaling might be involved in responses to other types of physical interaction between plant species and their natural surroundings. Takahashi et al. (2011) further defined H2O2-based signaling pathways by establishing connections between typical plant responses to abiotic stressors and responses to wounding events. In Arabidopsis, a mitogen-activated protein kinase 8 (MPK8) was shown to connect protein phosphorylation, calcium, and ROS in a wound-signaling pathway. Firstly, mechanical wounding of the plant triggered an increase in cytosolic calcium in local cells, which promoted the binding of calmodulins and the subsequent formation of active Ca2+/CaM complexes. These complexes then activated MPK8, which was also phosphorylated and activated by a MAPKK MKK3 in the typical MAPK cascade. Figure 7 provides a visual summary of this calcium dependent pathway that begins with a typical herbivore attack. Takahashi et al. 2011 then considered the effect of activated MPK8 on expression levels of three major molecular markers for wounding and H2O2-response: RbohD, zinc finger protein-encoding Zat12, and the serine/threonine kinase-encoding OXI1. In wild-type plants, they observed induction of expression of all three markers; however, in mutant mpk8 species they observed a relative increase in this induction. This precise MAPK pathway negatively regulates buildup of reactive species such as hydrogen peroxide. It is important to note the discrepancy between this finding and the previously described involvement of MAPK in positively facilitating responses to environmental stressors such as cold temperatures and osmotic stress. Other important enzymes have been shown to be involved in H2O2-stimulated responses as well. In Arabidopsis, the enzyme glutathione reductase (GR) is responsible for the maintenance of the major cellular thiol glutathione in a reduced state (Mhamdi et al. 2010). Within this group, the gene GR1 encodes the enzyme. Knocking out GRI resulted in predominantly oxidized glutathione (GSSG), as indicated by a decreased percentage of glutathione reduction that is shown in Figure 8. This version of the compound found in the mutants blocked several of the well-known pathways needed for wound and defense responses in the plant. The oxidized status of the thiol inhibited normal H2O2–mediated physiological responses following a stress event including the limitation of lesion formation, accumulation of salicylic acid, induction of pathogenesis-related genes, and signaling through jasmonic acid pathways. Knocking out GR1 also had a supplementary effect of increasing the number of transcripts of glutathione-associated genes, including two phi classes (GSTF2, GSTF8) and three tau class (GSTU7, GSTU8, GSTU19) enzymes that are involved in peroxidase function (Figure 9). Shu et al. (2011) examined a different plant species, but also discovered consistent findings. In the tomato plant Lycopersicon esculentum, knocking out the glutathione reductase-encoding gene (LeGR) resulted in greater initial accumulation of hydrogen peroxide, lower photosynthetic rate, and more cotyledon chlorophyll loss compared to controls (Shu et al. 2011). The loss of GR function was also correlated with a decrease in the activity of an ascorbate peroxidase, accounting for the heightened H2O2 levels. Therefore, glutathione reductase might play a central role in stimulating some of the early biochemical pathways that are necessary for plants to respond to stressors in their surrounding environment. Alteration of these pathways by the absence of the GR, in turn, can impact steps in the signal transduction cascades that directly or indirectly involve hydrogen peroxide. Figure 6 – Relative MtRboh gene expression (a) and nitrogen fixation activity (b) resulting from RNAi of MtRbohA in Medicago truncatula.
Figure 6a compares expression of seven identified putative RBOH-encoding genes in the Medicago genome between wild-type controls (functionally expressed MtRbohA) and plants resulting from RNAi of MtRbohA. Figure 6b compares ARA (acetylene reduction assay)—a measure of nitrogen fixation activity--- between wild-type controls and RNAi plants. Light grey bars represent controls, while black bars represent RNAi plants. * indicates significant difference compared to controls, p < 0.05. Errors bars +/- SE (Modified from Marino et al. 2011) Figure 7 – Calcium dependent pathway for activation of MPK8, as a response to herbivory.
Schematic representation of a mitogen-activated protein kinase 8 (MPK8) and how it connects to protein phosphorylation, calcium, and ROS in a wound-signaling pathway that begins with herbivore attack and ends with ROS production. (Modified from Tena et al. 2011) Figure 8- Effects of knockout of the GR1 gene in Arabidopsis.
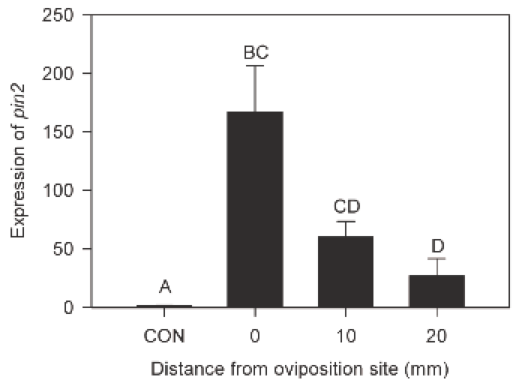
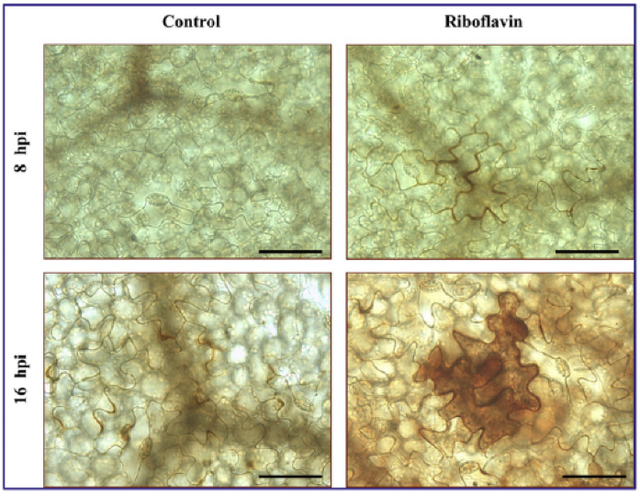
gr1 mutants across many conditions (short day SD, high CO2, long day LD) are compared to a control Arabidopsis plant used (Col-0), on the parameter “Glutathione reduction state” (% GSH). Cat2 is another mutant that was compared in the study to the control and gr1 mutants. (Modified from Mhamdi et al. 2010) Figure 9- Induction and repression of specific glutathione-associated gene transcripts following GR1 knockout in Arabidopsis. Induction and repression are measured in relation to log2 transcripts on the y-axis (which is a relative measure to the control Arabidopsis plant Col-0). Positive, upward graphs indicate induction, while negative, downward graphs indicate repression. The first two rows are dedicated towards induction effects, while the last is dedicated towards repression effects. (Modified from Mhamdi et al. 2010) As a complement to several endogenous compounds, many external factors can influence the strength of defense responses in plants. Petzold-Maxwell et al. (2011) revealed that plants have defense mechanisms to respond not only to herbivory but also to insect oviposition, such as a hypersensitive response or necrosis resulting in desiccation of insect eggs. However, there has been an expansion in focus from plant mechanisms that destroy eggs placed on the plant to mechanisms that impact the survival potential of neonates or insect larvae. Kim et al. (2012) examined whether the tomato plant Solanum lycopersicum showed a defense response to oviposition by the tomato fruitworm moth Helicoverpa zea, and subsequently detected significant presence of H2O2 upon treatment. Simultaneously, the transcriptional level of protease inhibitor2-encoding pin2 gene that resulted from oviposition was measured, as a way to represent the plant’s preemptive defense against larvae (the protease does not impact eggs, but rather targets live, feeding insects). Kim et al. (2012) discovered that expression of pin2 was significantly induced, especially at sites closer to the location of initial oviposition. This is shown by Figure 10, which indicates that the expression levels of the gene decrease as distance from the site of oviposition increases (Kim et al. 2012). Different plant and insect species also demonstrate a simultaneous increase in presence of H2O2 and transcription of pin2 in the plant (Puysseleyr et al. 2011). Therefore, it appears that insect eggs can stimulate defense mechanisms in various plants that are mediated by hydrogen peroxide and are similar to wound-induced signaling events. Treatment with different micronutrients can also have a modulating effect on H2O2- mediated defense pathways in plants. Exogenous application of riboflavin at concentrations ranging from 10-1000 µm on bean plants infected with Botrytis cinerea resulted in a 25% reduction in lesions relative to control plants (Azami-Sardooei et al. 2010). Early accumulation of hydrogen peroxide was also detected in these riboflavin-treated specimens compared to controls, approximately four hours post-infection (Figure 11). Increased expression and activation of lipoxygenase was also observed, along with subsequent pest-resistance pathways stimulated by the enzyme. When the same methodology and tests were applied to tomato plants, hydrogen peroxide accumulation was not observed until forty-eight hours post-infection and stimulation of lipoxygenase activity by riboflavin was not detected (Azami-Sardooei et al. 2010). Some micronutrients can act as stressors themselves. Cadmium is one non-essential metal that has been shown to inhibit root growth and disrupt cellular metabolism in plants. Tamas et al. (2009) found that when cadmium was applied to the longitudinal axis of barley root tips, hydrogen peroxide production was localized to and elevated in the root elongation zone and the differentiation zone of the plant. The heightened level of the compound was suggestive of many processes involved in root growth inhibition, including cross-linking of cell walls and lignification. Taken as a whole, these studies suggest that specific micronutrients can exert specific effects on specific plants, either as protective reinforcers of natural responses or as defense-inducing stimuli themselves. Finally, Maruta et al. (2012) explored the ironic need for plants to hold specific protective mechanisms against potential damage to cellular structures from endogenous accumulation of hydrogen peroxide during stress and defense responses. Of significant importance was the gene encoding the cytosolic ascorbate peroxidase 1 in Arabidopsis, called APX1, within the context of a wounding event. This enzyme is a crucial regulator of the redox state and ROS levels in plants, including during the absence of environmental stressors. Maruta et al. (2012) compared the impact of wounding with scissors on hydrogen peroxide levels in wild-type plants and in mutants whose APX1 was knocked-out. A heightened sensitivity to wounding in the mutants was observed, as demonstrated by the much more widespread accumulation of H2O2 in undamaged regions relative to the wild-type phenotype shown in Figure 12. Furthermore, significantly higher oxidative damage in the chloroplasts and nucleus of mutant leaves were noted. Figure 10 – Strength of induction of pin2 with distance from oviposition site on leaf of Solanum lycopersicum.
A graph indicating the relative levels of expression of pin2 at distances of 0, 10, and 20 mm from the oviposition site on a leaf of Solanum lycopersicum (eggs deposited by Helicoverpa zea). CON represents control plants with no oviposition occurring on them. Letters above bars indicate significant difference; Chi-square = 14.4695, p = 0.0023 (From Kim et al. 2012) Figure 11 – Effect of riboflavin on hydrogen peroxide accumulation in bean epidermal cells, four hours post-infection with B. cinerea.
The image on the left shows the effects of hydrogen peroxide accumulation following infection with B. cinerea, where no riboflavin was applied exogenously (control). The image on the right shows the effects of hydrogen peroxide accumulation following B. cinerea introduction, where riboflavin was applied exogenously. Treatment (control v. riboflavin) was given at both 8 and 16 hours post infection, as indicated. (Modified from Azami-Sardooei et al. 2010) Figure 12 – Accumulation of hydrogen peroxide and phenotypic effect of wounding Arabidopsis.
The image on the left shows the effects of hydrogen peroxide accumulation following wounding (with scissors) on a wild-type Arabidopsis with a functionally expressed APX1 (for a cytosolic ascorbate peroxidase 1). The image on the right shows the effects of hydrogen peroxide accumulation following wounding (with scissors) on a knockout-APX1 mutant. Distal site refers to an area separate from the original site of the wound. Wounded site refers to area where wound was inflicted. (Modified from Maruta et al. 2012) Figure 13- Functional classification and distribution of 24 proteins with altered expression in rice plant mutants lacking APX1 expression.
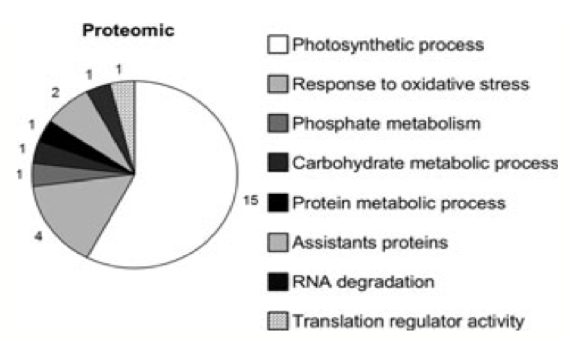
Pie chart representing the various functions of 24 induced genes in the rice plant transcriptome (set of all RNA molecules produced). Each slice is represented by a specific pattern, as noted by the key to the right of the chart. (From Ribeiro et al. 2012) Despite these findings in experiments using Arabidopsis, it is important to note that some plant species have yet another defense mechanism to counter the loss of APX1 expression—modulating other genes involved in metabolic pathways. Ribeiro et al. (2012) demonstrated how knockout of OsAPX1 and OsAPX2 in rice plants resulted in normal phenotype and development similar to wild-type controls, despite an accumulation of hydrogen peroxide. Proteomic analysis and mass spectroscopy were used to pinpoint differentially expressed proteins in the mutants, revealing twenty-four proteins corresponding to genes showing increased expression. Of these proteins, sixty-four percent was associated with photosynthesis functions (Figure 13). There was an upregulation of genes involved in the electron flux of the chloroplast electron transport chain, including those encoding plastocyanin and ferredoxin. An increased expression of antioxidant-encoding genes was also noted, including the peroxiredoxin and glutathione-S-transferase genes. Overall, these changes in gene expression can serve as a holistic compensatory mechanism to protect the plant from physical damage resulting from loss of peroxidase function, ensuring that cells receive sufficient nutrients and compounds to safeguard key organelles.Continued on Next Page » Suggested Reading from Inquiries Journal
Inquiries Journal provides undergraduate and graduate students around the world a platform for the wide dissemination of academic work over a range of core disciplines. Representing the work of students from hundreds of institutions around the globe, Inquiries Journal's large database of academic articles is completely free. Learn more | Blog | Submit Latest in Biology |