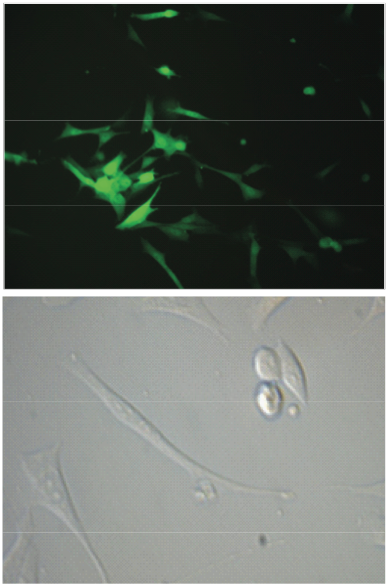
From Discussions VOL. 5 NO. 2Development of a Micro-integration Technique for the Fabrication of a Cell-Fiber Scaffold Complex in Heart Valve Tissue EngineeringResultsInitial studies were carried out using a single polymer/cell types construct: (PCl)/(GFP+ 3T3 Fibroblasts) . Live and functioning cells spread themselves out on a surface in the attaching process. Microscopic images of the cells in the electrospun PCl sample taken at 1, 3, 7, 10 and 14 days after initial seeding showed increasing cell diameter, which suggests excellent cell attachment and growth. Visualization of attaching cells is shown in Figure 2 (right). A live/dead fluorescent stain performed on these cells confirmed cell viability and proliferation as illus-trated in Figure 3. The green florescence indicates living cells, while the red fluorescence shows the amount of dead cells present in the sample. The image shown in Figure 2 illustrates the presence of a majority of green fluorescently stained cells. This result provided further evidence for cell viability and proliferation. To determine statistically the number of live cells in the sample after the initial 24 hour period, dead cells were washed away with PBS, and the remaining alive ones were counted manually. Cell counts data were taken from 10 equally sized areas within the scaffold. Subsequent calculation of live and dead cell averages and deviations showed approximately 80% cell viability in comparison to the total number of cells counted before the electro-spinning/spraying process. In addition, even number of live and dead cell counts determined at different thicknesses and widths of the scaffold also demonstrated uniform cell densities and cell distribution via the micro-integration process. Figure 4 (next page) presents visual images of homogenous cells distributed throughout the matrix taken by light and fluorescence microscopy. Furthermore, optical and scanning microscopy images of a portion of seeded scaffold fixed in paraformaldehyde indicated improved cell growth and adhesion on fibrous surfaces with small pores, in comparison to a smooth film of the same polymer. These data were obtained by comparing cell growth profiles on a mat of electrospun PCl versus a PCl cast film. Subsequent experiments involving the endothelial progenitor cells and the fibroblasts seeded in layers of electrospun collagen and PCl also showed good cell viability and uniform cellular distribution. Histological sectioning and staining of the samples are still in progress. Figure 2: fluorescence and light microscopy of alive and well-attaching GPF+ 3T3 fibroblasts before electrostatic seeding.
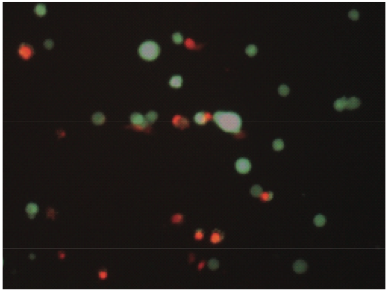
Figure 3: Image of 3T3 fibroblast/PCl sample stained with the alive/dead fluorescence assay. Green indicates live cells and red indicates dead cell.
DiscussionIn these experiments, an innovative approach involving simultaneous electrospinning of polymer and electrospraying of cells was successfully developed and optimized in seeding of the GFP+ 3T3 fibroblasts and the progenitor cells in the electrospun fibrous scaffolds. Cell viability and proliferation was maintained both through the electrospinning process and subsequent culturing of the constructs. Cells were also found to be uniformly distributed throughout the cross section of the scaffold samples. The optimiza-tion of the electrospin/spray process was achieved through varying the electro-spinning processing parameters, such as the flow rate of the polymer solution and the distance between the syringe and the drum. Varied fiber diameter and pore sizes were also obtained by controlling these parameters. During the micro-integration process, we overcame the difficulty in keeping cells alive during the two hour electrospray process by constantly spraying media onto the developing cell/fiber construct. We also coated the fibers with laminin to promote better cell adhesion to the synthetic components of the scaffold. Upon fabrication of the cell-seeded scaffolds, the unfixed sample portion cultured in cell media showed substantial viability and differentiation over time. However, we believe that we can induce further cell proliferation by adding growth factors. Data on fixed samples processed for histological examination with hematoxylin and eosin staining to identify cell location within the scaffold upon initial seeding will be obtained in the next set of experiments. Also, maturation of seeded cells from progenitor forms will be assessed based on their genes and protein products by multiplex protein analysis and real-time PCR analysis. The extent of cell differentiation from precursor to mature phenotype induced by the scaffold with or without growth factor delivery must be determined in order to demonstrate valve-like histological cell layering and cell type matched matrix secretion. Figure 4: Light and fluorescence microscopy of electrospun poly(caprolactone) scaffolds seeded with green fluorescent protein expressing 3T3 fibroblasts via electrostatic spraying, showing the uniform distribution of cells throughout the matrix.
ConclusionThe expected outcome of these experiments showed that 1) cells survived the electrostatic seeding process and 2) cells adhered to the fibers and were viable and multiplying within the scaffold. Uniform distribution of cells within the scaffolds also indicated successful application of the micro-integration process. These preliminary investigations showed promising results in the development of both a multifunctional scaffold and a state-of-the-art methodology for tissue engineering scaffold optimization, which will lay a strong foundation for future work towards generating a fully functional heart valve substitute. In future experiments, several other polymers will be considered for scaffolding, either on their own or as part of a multi-polymer scaffold. These polymers include, but are not limited to, synthetic biocompatible materials such as PLGA, PGA, poly-4-hydroxybutyrate, and several poly(urethanes). Natural polymers such as collagen, elastin, and fibrinogen will also be investigated for incorporation with the synthetic polymers in an attempt to better mimic the native cellular architecture and the mechanical properties of the native heart valve tissue. The next stages of work will concentrate on producing a tri-layer scaffold with endothelial precursor layers on the top and bottom, sandwiching a center layer of fibroblast progenitor cells, and determining the biocompatibility and hemodynamic function of the engineered valve in a bioreactor system, prior to valve implantation and testing in mice models. AcknowledgmentsThe effective collaboration and cooperation among all members of our interdisciplinary team have made this work successful. Specifically, I would like to thank Dr. Gary Wnek for his guidance and expertise in polymer science, electro-spinning and biomaterials, as well as providing access to polymer processing facilities at Case Western Reserve University Department of Macromolecular Science and Engineering. I would also like to thank Dr. Yakov Elgudin and Dr. Peter Havernik from the Case Western Reserve University School of Medicine for provid- ing considerable expertise in heart valve tissue engineering, tissue culture, delivery systems, cell biology and histology. I must also thank Meghan Smith for her patience, super- vision and invaluable assistance in carrying out lab procedures and techniques. Lastly, I want to thank everyone else for their support in making this research project possible. ReferencesBoland, E.D., et al. "Tailoring tissue engineering scaffolds using electrostatic processing techniques: a study of poly(glycolic acid) electrospinning." Journal of Macromolecular SciencePure and Applied Chemistry 38 (2001): 1231-1243. Boudroit, U., et al. "Electrospinning approaches toward scaffold engineering A brief overview." Artificial Organs 30.10 (2006): 785-792. Brody, S. and Pandit, A. "Approaches to Heart Valve Tissue Engineering." Journal of Biomedical Materials Research Part B: Applied Biomaterials (2007): 16-43. Fong, P., et al. "The use of polymer based scaffolds in tissue-engineering heart valves." Progress in Pediatric Cardiology 21 (2006): 193-199. G. L. Bowlin, K. J. Pawlowski, E. Boland, D. G. Simpson, J. B. Fenn, G. E. Wnek and J. D. Stitzel, "Electrospinning of Polymer Scaffolds for Tissue Engineering," Tissue Engineering and Biodegradable Equivalents: Scientific and Clinical Applications, K. Lewandrowsky, D. J. Trantolo, J. D. Gresser, M. J. Yaszemski, D. E. Altobelli and D. L. Wise, Editors, Marcel Dekker, Ch. 9, (2002): pp. 165-178. Hohman, M.M., et al. "Electrospinning and electrically forced jets. I. Stability theory." Physics of Fluids 13 (2001): 22012220. Hsu, C.M. and Shivkumar, S. "N,N-dimethylformamide additions to the solution for the electrospinning of poly(e-caprolactone) nanofibers." Macromolecular Materials and Engineering 289 (2004): 334-340. Li, Y, et al. "Effects of filtration seeding on cell density, spatial distribution, and proliferation in nonwoven fibrous matrices." Biotechnology Progress 17 (2001): 935-944. Matthews, J.A., et al. "Electrospinning of collagen nanofibers." Biomacromolecules 3 (2002): 232-238. Mendelson, K. and Schoen, F.J.. "Heart Valve Tissue Engineering: Concepts, Approaches, Progress, and Challenges." Annals of Biomedical Engineering 24.12 (2006): 1799-1819. Neuenschwander, S. and Hoerstrup, S.P.. "Heart valve tissue engineering." Transplant Immunology 12 (2004): 359-365. Nkomo VT, Gardin JM, Skelton TN, et al. Burden of valvular diseases: a population-based study. The Lancet Online. August 18, 2006; Vol 368; pp 1005-1011. Sill, T.J. and von Recum, H.A.. "Electrospinning: Applications in drug delivery and tissue engineering." Biomaterials 29 (2008): 1989-2006. Sodian, R., et al. "Evaluation of biodegradable three dimensional matrices for tissue engineering of heart valves." American Society of Artificial Internal Organs Journal 46 (2000): 107-110. Zund, G., et al. "The in vitro construction of a tissue engineered bioprosthetic heart valve." European Journal of Cardiothoracic Surgery 23 (1997): 493-497. Suggested Reading from Inquiries Journal
Inquiries Journal provides undergraduate and graduate students around the world a platform for the wide dissemination of academic work over a range of core disciplines. Representing the work of students from hundreds of institutions around the globe, Inquiries Journal's large database of academic articles is completely free. Learn more | Blog | Submit Latest in Health Science |