An Assessment of the Origins and Culpability of the Opioid Crisis in the United States
By
2022, Vol. 14 No. 03 | pg. 1/1
IN THIS ARTICLE
KEYWORDS
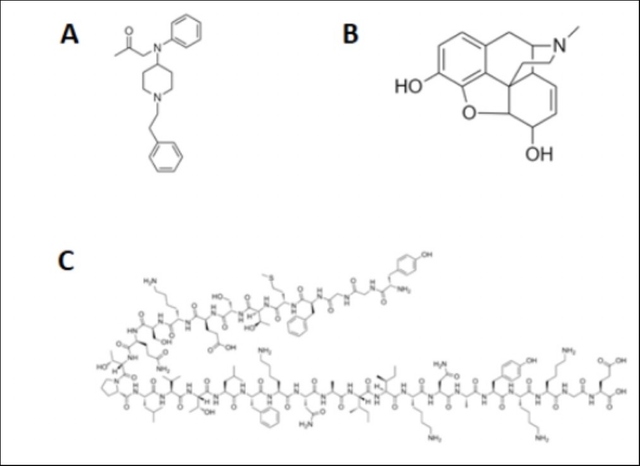
AbstractThe use of synthetic opioids in the United States in the past 30 years has created an epidemic the likes of which our healthcare and law enforcement systems have never before encountered. Although some opioid analogs, like fentanyl, were developed with the intention of managing chronic pain, these potent drugs have, through their addictive qualities, trapped millions of Americans in a deadly cycle of dependency. This review addresses the major routes by which synthetic opioids are illegally shipped into the United States, where they are then sold to the IntroductionPrescribed opioids are a family of compounds intended to treat moderate to severe pain ranging from post-trauma surgery relief to mild back pain. Toward the end of the 20th century, opioid prescriptions dramatically rose in prevalence in the United States; in 1990, doctors prescribed two to three million opioids per year. By 1999, this number had jumped radically to eleven million (Rummans et al., 2018). As the opioid epidemic progressed, advances in science and medicine contributed to synthesizing more potent opioid analogs such as fentanyl, a newer synthetic opioid that is far more potent than morphine. Consequently, fentanyl is intended for individuals who have developed a tolerance to weaker analogs or have severe chronic pain. Drug manufacturers marketed their new and improved drugs to physicians by providing evidence that they were less addictive than those already prescribed to manage pain for similar conditions (Haffajee & Mello, 2017). However, as these medications became more common, patients developed a physical dependence on the drugs, and thus, what we know today as the opioid crisis began.Mechanism of Action of OpiatesOpiate ReceptorsOpioid receptors are responsible for many different physiological reactions to opioid ligands within the body. These reactions can include cell proliferation, emotional response, epileptic seizures, immune function, feeding, respiratory and cardiovascular control, neurodegenerative disorders, and other responses (Feng et al., 2012). Because these receptors have such a wide area of effect, any increase or decrease of ligand concentration can lead to intense physiological responses throughout the entire body. According to Boom et al (2012), one of the main opioid receptors in the body is the μ-opioid receptors in the brain. These receptors play a role in regulating pain, but as a side-effect of opioid agonists like morphine and others, they suppress respiratory function. Physiological EffectsWhen morphine binds to these receptors and dopamine is released in greater quantities than under normal circumstances, the most notable effects are observed. Because dopamine is one of the primary neurotransmitters of the mesolimbic system and the central reward center of the brain, it affects many processes of the body (Girault & Greengard, 2004; Samaha et al., 2021). One neural pathway includes the ventral tegmental area and nucleus accumbens circuit (VTA-NAc) (Kauer & Malenka, 2007). VTA-NAc releases dopamine in response to favorable stimuli (e.g., food when hungry). When opioids are introduced into the body, the dopamine-mediated cellular response results in desiring a repeated stimulus signal, and addiction begins. Figure 1. Structures of common opioids fentanyl (A) and morphine (B) compared to beta-endorphin (C) (Pasternak & Pan, 2013). The dopamine response to opiates can also lead to a state of euphoria, pain relief, confusion, and slurred speech (Nestler, 1996; Wise & Bozarth, 1985). These opioid effects persist for more extended periods than their actual presence within the body, a contributing factor to their harm. More specifically, heroin's half-life is under 30 minutes, but the consequences continue for 3-5 hours after ingestion (Jenkins et al., 1994). Opioid harm to people that abuse these drugs (Table 1) is because we can become tolerant to the drug, and we take more and more over time. Due to damage to the liver and kidneys, opioids cause dehydration, hypotension, urinary retention, and muscle degradation. The cardiovascular system and its regulation are also greatly affected. Respiratory depression is a significant side effect that is induced by a μ-opioid response and can lead to brain and heart damage because these organs require large amounts of oxygen (Boom et al., 2012) . These effects show how multiple body systems can be affected by opioid abuse. The use of heroin taken intravenously or intramuscularly can result in an infection of the skin, heart, or lungs (Milroy & Parai, 2011). Furthermore, tolerance, the building up of effects that reduces the body response, leads to higher doses being required for the same effect. This increase leads to more adverse cardiovascular effects, such as clogging of blood vessels, rerouting veins, amenorrhea in women, endocrine dysfunction, and depression (Seyfried & Hester, 2012). These factors are some reasons why opioids should never be taken recreationally and only used under close supervision with a specific medical purpose. Table 1. Long-term Effects of Heroin
Heroin, an opium derivative, is identical to morphine except for two acetyl groups (Figure 1). These acetyl groups make heroin more lipid-soluble in blood than morphine, allowing faster and more efficient blood-brain barrier penetration (BBB). Although heroin effectively becomes morphine after deacetylation, the faster penetration translates into higher potency. Fentanyl has a more rapid effect than heroin because fentanyl has less rigid fused ringed structures (Figure 1) (Vardanyan & Hruby, 2014). After crossing the BBB, these drugs then attach to μ-opioid receptors in the brain. The endogenous opiates that generally bind to μ-opioid receptors are much larger molecules such as enkephalins, beta-endorphins, and dynorphins (Pasternak & Pan, 2013). Morphine possesses the capacity to bind to these receptors and cause a dopamine release that otherwise would not have occurred. The Supply LineAlthough the exact numbers are in dispute, the drug trade is estimated to be a $320 billion business (Insulza, 2013). Since the drug trade is a large business, it is best to follow a model in which responsibility is apportioned between the key aspects of the supply line, from manufacture to distribution. HeroinAccording to Sneader (1998), heroin was first commercially synthesized by Felix Hoffmann at Bayer Pharmaceutical, in which it was trademarked as “heroin.” Although Bayer believed it to be a non-addictive morphine substitute, addictive properties were soon discovered, and the Harrison Narcotics Act first targeted heroin in 1914, which regulated heroin production and distribution in the United States. The general production of heroin starts with the growth and harvest of opium flower seed pods, extraction of opium from the seed pod, separation of morphine from opium, creation of morphine base, production of heroin from morphine, and the cutting of heroin (Marciano et al., 2018). The opium sap is mixed with boiling water and lime and morphine is extracted. It is then boiled and repeatedly filtered until it has been converted into a brown paste known as morphine base. Morphine base is then combined with other chemicals that convert opium into heroin. Most heroin manufacturers are not professional chemists, so their yields are either impure due to a lack of skill or are deliberately impure so that cheaper or more accessible substances can be used to make a greater profit. Cutting agents include sugar, caffeine, flour, quinine, acetaminophen, and fentanyl. Opium is a problematic plant to grow because of high temperature and humidity requirements. These requirements, combined with the fact that yield from the opium seed is low, limit major growing areas to those such as the Golden Crescent and the Golden Triangle located in Western and Southern Asia, respectively (Dorabjee, 2015; Farooq et al., 2017). Once grown, the morphine has to be processed into heroin. Heroin factories are large and conspicuous, so they are often constructed in isolated areas, such as the hills of Sicily (by the famous French Connection of the Sicilian Mafia), the Burmese rainforests (by the many independence and guerrilla organizations), or the Mexican desserts (by the many drug cartels) where these facilities can be easily concealed (Ciccarone, 2009). After processing, heroin is then sent to a distributor (Agar & Reisinger, 2002). The Burmese have traditionally used Chinese (mainland and Taiwanese) distributors, who send them to Triads. These Chinese gangsters have existing routes to the Americas through human trafficking. The Mexicans use low-level members of the cartels living in the United States. The Mafia favors Unione Corse, French criminal syndicates, and their allies in the Italian-American Mafia. The distributors then recruit local gangs and associates to sell the drug to the consumer. The traditional approach by the Drug Enforcement Administration (DEA) has been to track down the source and slow the flow of precursor chemicals. Until the 2010s, heroin sales and deaths generally decreased for the first time since 1990 (Westhoff, 2019). This approach has largely been met with success in terms of heroin, except for Burmese guerrilla movements. FentanylBelgian physician Paul Janssen first invented fentanyl in 1960 through legitimate methods, but it spread to other illegal avenues as early as the 1970s. Fentanyl abuse did not become a major problem until the 2010s during the decrease of heroin supply. This demand has prompted new drug development methods, which follow the pattern as mentioned earlier with a few key differences (Westhoff, 2019). The dominant precursor chemical of fentanyl is piperidine, obtained from black pepper. Piperidine is surrounded by two phenyl groups and a propanamide group and can be modified in many ways to generate numerous fentanyl-like molecules (Vardanyan, 2014). Manufacturers of designer drugs, drugs that are analogs of existing drugs, are professional chemists (chemists with college degrees as opposed to street chemists). Most of them pull their recipes from scientific papers about fentanyl research. Some of these papers have instructions to make fentanyl analogs. Even if only the structures are known, the syntheses may not be challenging to figure out (Westhoff, 2019). The above synthesis pathways were found within five minutes of browser searching. Most of these drugs come from mainland China. The combination of vast amounts of capital, low regulation, cheap labor, and government corruption have allowed hundreds of chemical companies to rise and flood the market. According to Westhoff (2019, Fourteen, para. 16), "They can be broken down into three categories: companies that make drugs that are not just illegal in Western countries but illegal in China…, companies that specialize in [new psychoactive substances] that are illegal in the West but…just legal in China…, and companies that…focus on…fentanyl precursors and anabolic steroids that illegal in Western countries but legal in China". The first two categories often operate openly in China, with large factories, public company headquarters, and open communications channels that are legal. The third category ships it to a factory in some other country, such as Mexico. Mexico's long history of heroin processing means that they have the chemical knowledge to convert the precursors into fentanyl. These factors make Mexico a popular shipping destination for many Chinese factories (Westhoff, 2019). China may be responsible for up to 90% of the Mexican drug cartels' fentanyl precursors. The international scope of the precursor and drug production has frustrated law enforcement officials and thrown out the traditional War on Drugs methods, so much so that the Drug Enforcement Administration has largely side-stepped the issue (Westhoff, 2019). The DEA has effectively no contacts in China, where many of these precursors are made and are mostly importing equipment to Myanmar rather than actively coordinating drug policy. Their involvement in Mexico has been largely ineffective, with an ever-increasing number of cartels and scale of violence since the beginning of DEA's involvement in that country. As a result, the DEA is pushing for a domestic focus, which has been met with little success, based on the rapidly increasing number of opioid users in the U.S. since the 1990s. Suggested Culpability for the Opioid CrisisThe parties responsible for the opioid crisis must be identified to resolve the problem. This paper is an opinion on the apportion blame to all parties that can be made accountable, based on three factors:
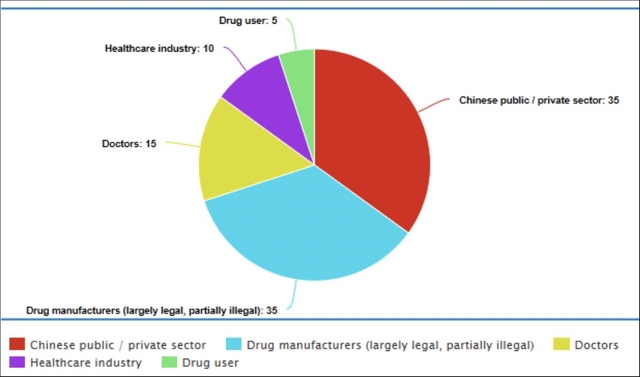
Drug companies deserve a sizable portion of the blame. Most of the attention regarding accountability has been on companies like Purdue Pharma, who created OxyContin, and then sought to market it to doctors and hospitals as an ideal way to deal with patient pain (Jayawant & Balkrishnan, 2005). Purdue Pharma even touted this new drug as less likely to be addictive, although there was little to no evidence to back up this claim (Aquina et al., 2009; Chow, 2019; Zee, 2009). This advertising campaign was wildly successful and effectively created an artificially elevated demand from doctors who wanted to reduce addiction rates (Baumrucker, 2001; Zee, 2009). However, beyond creating OxyContin, Purdue Pharma has had a more considerable impact on the opioid crisis in that it started a culture of prescribing, where doctors were pressed to give painkillers to patients who did not necessarily need them (Marks, 2020). This culture of prescribing is effectively the construction of a new, previously nonexistent demand chain by drug manufacturers. Purdue Pharma deserves a large portion of the blame due to the intentionality and apparent singular motivating force for selling more drugs. It should also be noted that there are sizable illegal drug manufacturers that play a role in the supply chain. However, illicit drug manufacturers emerged after the rise of the large pharmaceutical market and illegal drug distributors. Although they exacerbate the crisis by making multiple new supplies, they have not started (Westhoff, 2019). Because the combination of legal and illegal drug manufacturers forms a collective positive feedback loop of higher drug production, they must be judged collectively. Altogether, 35% of the blame for the opioid crisis is placed on them, with a stronger emphasis on legal manufacturers such as Purdue Pharma. Doctors are not without blame for the opioid crisis as well. In their goal to reduce patient suffering, doctors often inadvertently overprescribe opioids. An estimated 80% of those who try heroin are first introduced to opiates through prescription medications (Cicero et al., 2014). As mentioned, the overprescribing that has taken place over the past 20 years is partly due to the misinformation, intentional or not, issued by pharmaceutical companies. It also appears that doctors were monetarily incentivized to prescribe their opiates. Instead of prescribing opioids solely to treat acute pain, doctors began to prescribe opioids for chronic pain, a practice that is still common (Nuckols et al., 2014). As far as intentions go, doctors deserve relatively low blame because their role in the opioid crisis often comes from trying to prevent patient suffering. Still, doctors certainly deserve at least a sizable portion of the responsibility for both preventability and burden of action. Our healthcare system is set up with checks and balances to prevent systemic disasters like this one, and doctors were supposed to be one of those checks. It is a logical conclusion that if doctors had not overprescribed opioids, theme addiction could not start as frequently from prescription medication. In general, doctors have moved through four years of medical school and several years of training afterward to be experts on patient health. Subsequently, they have been given enormous responsibility for deciding who does and does not need opioids and how long they are needed. Whether through lack of specific drug-based education, laziness, overwork, or even greed, it is inescapable that some doctors have failed to protect their patients, and for these reasons, 15% of the blame has been placed on them. The next party is the public and private sectors of China. The Chinese government promotes low regulation to stimulate growth, and the Chinese private industry exploits these weaknesses to become the primary origin site for fentanyl and similar opioids (Westhoff, 2019). These large corporations profit from the opioid crisis. China gets this particular attention because of the unique integration of its government and economy. Much like homegrown manufacturers, their motivation is profit. They must take some blame for preventability. Because they do not background check foreign buyers, they often knowingly sell to distributors instead of addicts (Westhoff, 2019). Although they have obligations under international drug trafficking treaties, the treaties are vague, and ultimately the crisis is not affecting China as severely as the United States. For these reasons, China is allotted 35% of the blame. The United States healthcare system itself must also take part of the blame. The money-driven nature of healthcare in the United States opened the door for drug companies to take advantage of the system by profiting from introducing new opioids into the market with few consequences for these companies. The nature of the U.S. healthcare system has also created an environment where painkillers are almost always going to be a more prominent source of pain management treatment due to their cheaper cost. Often, insurance companies are unwilling to pay for alternative methods of controlling pain, resulting in a much larger reliance on opioids (Blum et al., 2018; Bonakdar et al., 2019). Failure to recognize these obstacles and develop a system to prevent large-scale opioid addiction is seen as a failure by the U.S. healthcare system. The U.S. healthcare system as a whole should have been capable of stopping this crisis in a variety of ways, including better training for healthcare professionals to recognize when opioids are not needed and ways to combat chronic pain that do not involve addictive medications. Only recently does it appear the healthcare system is starting to realize the totality of this crisis, too late for thousands of Americans. The amount of blame given to the U.S. healthcare system is 15%. The final component in this apportioning of blame is the individual drug users themselves. Political circles that fund drug enforcement are split. One group blames the individual for physically taking the drug, the logic being that no one is forcing them to consume the drug, so they are an agent of free will. The other group states the individual has a series of developed or congenital traits that make them susceptible to drugs, so they have predispositions that make it more difficult for them to act as an agent of free will. The addictive personality consists of the following traits: impulsivity, sensation-seeking, nonconformity, weak commitment to achievement, social alienation, toleration for deviance, heightened stress, and a lack of coping skills (Nelson, 1983). There is also some evidence for a genetically-based behavior disposition. The issue with the idea of an addictive personality is that it is overbroad in its coverage. Practically everyone will experience heightened stress or be socially alienated at some point in their life. The other parameters, tolerating deviance, nonconformity, impulsivity, and sensation-seeking, are too vague to be used effectively, while only sensation-seeking is clearly defined. In combination with the addictive personality's origin from Alcoholics Anonymous and not being backed by scientific research (Szalavitz, 2015), the idea of a personality conducive to addiction must be discarded as a myth. However, genetic factors have been identified as conducive to addiction, with some genes modified to halt addictive impulses. While this research does not override the typical personality of an average human and the extensive side-effects following the initial high, the idea that it makes them non-culpable for their actions is dismissed. Considering all these factors, they are awarded 5% of the blame. We have chosen to acquit the drug enforcement agencies. Although the illegal drug market vacancy has allowed the explosion of the opioid crisis and the multi-source distribution of the drug supply line, the mere fact that this distribution has occurred shows that the agency's goals had been met. Additionally, the radical upheaval in the illegal drug market was largely unforeseeable. In summary, opioid overuse has a profoundly negative impact on human physiological functions. As a result, this abuse has led to thousands of deaths in the United States since pharmaceutical companies began a major push for opioid use for chronic pain in the 1990s. Many entities are to blame for letting the opioid crisis progress to this point, including doctors, the American healthcare system, and even individuals. But the lion's share of the blame belongs to drug manufacturers and China due to both their money-oriented motivations and their unwillingness to do more to fight this epidemic. This analysis contributes to the knowledge of the opioid crisis and will hopefully lead to better initiatives in fighting this substance abuse epidemic. ReferencesAgar, M., & Reisinger, H. S.. (2002). None. Culture, Medicine, and Psychiatry, 26(3), 371–396. https://doi.org/10.1023/a:1021261820808 Aquina, C. T., Marques-Baptista, A., Bridgeman, P., & Merlin, M. A. (2009). OxyContin abuse and overdose. Postgraduate medicine, 121(2), 163–167. https://doi.org/10.3810/pgm.2009.03.1988 Baumrucker S. J. (2001). OxyContin, the media, and law enforcement. The American journal of hospice & palliative care, 18(3), 154–156. https://doi.org/10.1177/104990910101800304 Blum, K., Jacobs, W., Modestino, E. J., DiNubile, N., Baron, D., McLaughlin, T., Siwicki, D., Elman, I., Moran, M., Braverman, E. R., Thanos, P. K., & Badgaiyan, R. D. (2018). Insurance Companies Fighting the Peer Review Empire without any Validity: the Case for Addiction and Pain Modalities in the face of an American Drug Epidemic. SEJ surgery and pain, 1(1), 1–11. Bonakdar, R., Palanker, D., & Sweeney, M. M.. (2019). Analysis of State Insurance Coverage for Nonpharmacologic Treatment of Low Back Pain as Recommended by the American College of Physicians Guidelines. Global Advances in Health and Medicine, 8, 216495611985562. https://doi.org/10.1177/2164956119855629 Boom, M., Niesters, M., Sarton, E., Aarts, L., Smith, T. W., & Dahan, A. (2012). Non-analgesic effects of opioids: opioid-induced respiratory depression. Current pharmaceutical design, 18(37), 5994–6004. https://doi.org/10.2174/138161212803582469 Chow R. Purdue Pharma and OxyContin – A commercial success but public health disaster. Harvard Public Health Review. 2021; 28. Ciccarone D. (2009). Heroin in brown, black and white: structural factors and medical consequences in the US heroin market. The International journal on drug policy, 20(3), 277–282. https://doi.org/10.1016/j.drugpo.2008.08.003 Cicero, T. J., Ellis, M. S., Surratt, H. L., & Kurtz, S. P.. (2014). The Changing Face of Heroin Use in the United States. JAMA Psychiatry, 71(7), 821. https://doi.org/10.1001/jamapsychiatry.2014.366 Dorabjee, J.. (2015). The view from the trenches. Harm Reduction Journal, 12(1). https://doi.org/10.1186/s12954-015-0078-6 Farooq, S. A., Rasooly, M. H., Abidi, S. H., Modjarrad, K., & Ali, S.. (2017). Opium trade and the spread of HIV in the Golden Crescent. Harm Reduction Journal, 14(1). https://doi.org/10.1186/s12954-017-0170-1 Feng, Y., He, X., Yang, Y., Chao, D., H. Lazarus, L., & Xia, Y.. (2012). Current Research on Opioid Receptor Function. Current Drug Targets, 13(2), 230–246. https://doi.org/10.2174/138945012799201612 Girault, J.-A., & Greengard, P.. (2004). The Neurobiology of Dopamine Signaling. Archives of Neurology, 61(5), 641. https://doi.org/10.1001/archneur.61.5.641 Haffajee, R. L., & Mello, M. M.. (2017). Drug Companies’ Liability for the Opioid Epidemic. New England Journal of Medicine, 377(24), 2301–2305. https://doi.org/10.1056/nejmp1710756 Insulza, José Miguel. (2013). The Drug Problem in the Americas: Studies. The economics of drug trafficking. Organization of American States. Jayawant, S. S., & Balkrishnan, R.. (2005). The controversy surrounding OxyContin abuse: issues and solutions. Therapeutics and Clinical Risk Management, 1(2), 77–82. https://doi.org/10.2147/tcrm.1.2.77.62911 Jenkins, A. J., Keenan, R. M., Henningfield, J. E., & Cone, E. J. (1994). Pharmacokinetics and pharmacodynamics of smoked heroin. Journal of analytical toxicology, 18(6), 317–330. https://doi.org/10.1093/jat/18.6.317 Kauer, J. A., & Malenka, R. C. (2007). Synaptic plasticity and addiction. Nature reviews. Neuroscience, 8(11), 844–858. https://doi.org/10.1038/nrn2234 Marciano, M. A., Panicker, S. X., Liddil, G. D., Lindgren, D., & Sweder, K. S.. (2018). Development of a Method to Extract Opium Poppy (Papaver somniferum L.) DNA from Heroin. Scientific Reports, 8(1). https://doi.org/10.1038/s41598-018-20996-9 Marks, J. H.. (2020). Lessons from Corporate Influence in the Opioid Epidemic: Toward a Norm of Separation. Journal of Bioethical Inquiry, 17(2), 173–189. https://doi.org/10.1007/s11673-020-09982-x Milroy, C. M., & Parai, J. L. (2011). The histopathology of drugs of abuse. Histopathology, 59(4), 579–593. https://doi.org/10.1111/j.1365-2559.2010.03728.x Nestler E. J. (1996). Under siege: The brain on opiates. Neuron, 16(5), 897–900. https://doi.org/10.1016/s0896-6273(00)80110-5 Nuckols, T. K., Anderson, L., Popescu, I., Diamant, A. L., Doyle, B., Di Capua, P., & Chou, R. (2014). Opioid prescribing: a systematic review and critical appraisal of guidelines for chronic pain. Annals of internal medicine, 160(1), 38–47. https://doi.org/10.7326/0003-4819-160-1-201401070-00732 Pasternak, G. W., & Pan, Y.-X.. (2013). Mu Opioids and Their Receptors: Evolution of a Concept. Pharmacological Reviews, 65(4), 1257–1317. https://doi.org/10.1124/pr.112.007138 Rummans, T. A., Burton, M. C., & Dawson, N. L.. (2018). How Good Intentions Contributed to Bad Outcomes: The Opioid Crisis. Mayo Clinic Proceedings, 93(3), 344–350. https://doi.org/10.1016/j.mayocp.2017.12.020 Samaha, A. N., Khoo, S. Y., Ferrario, C. R., & Robinson, T. E. (2021). Dopamine 'ups and downs' in addiction revisited. Trends in neurosciences, 44(7), 516–526. https://doi.org/10.1016/j.tins.2021.03.003 Seyfried, O., & Hester, J.. (2012). Opioids and endocrine dysfunction. British Journal of Pain, 6(1), 17–24. https://doi.org/10.1177/2049463712438299 Sneader W. (1998). The discovery of heroin. Lancet (London, England), 352(9141), 1697–1699. https://doi.org/10.1016/S0140-6736(98)07115-3 Vardanyan, R. S., & Hruby, V. J.. (2014). Fentanyl-related compounds and derivatives: current status and future prospects for pharmaceutical applications. Future Medicinal Chemistry, 6(4), 385–412. https://doi.org/10.4155/fmc.13.215 Ben Westhoff. (2019). Fentanyl, Inc. : How Rogue Chemists Are Creating the Deadliest Wave of the Opioid Epidemic: Vol. First edition. [E-book]. Atlantic Monthly Press. Wise, R. A., & Bozarth, M. A. (1985). Brain mechanisms of drug reward and euphoria. Psychiatric medicine, 3(4), 445–460. Van Zee, A.. (2009). The Promotion and Marketing of OxyContin: Commercial Triumph, Public Health Tragedy. American Journal of Public Health, 99(2), 221–227. https://doi.org/10.2105/ajph.2007.131714 Suggested Reading from Inquiries Journal
Inquiries Journal provides undergraduate and graduate students around the world a platform for the wide dissemination of academic work over a range of core disciplines. Representing the work of students from hundreds of institutions around the globe, Inquiries Journal's large database of academic articles is completely free. Learn more | Blog | Submit Latest in Health Science |