The Use of Ketamine as a Treatment for Depression and Alcohol Use Disorders
By
2020, Vol. 12 No. 10 | pg. 1/1
IN THIS ARTICLE
KEYWORDS
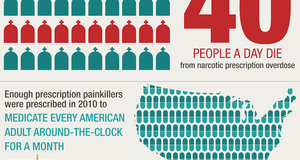
AbstractKetamine, described by the chemical formula C13H16ClNO, is most commonly associated with adolescent and adult recreational drug users and ravers who abuse this drug to experience a euphoric and dissociative state. Although this drug is a federal schedule III controlled substance as a result of this abuse potential, ketamine has experienced a renaissance in clinical interest in recent years.1 Although clinical use of ketamine is controversial, it has become a focal point of pharmacological research due to its considerably diverse molecular targets and neurophysiological properties.2 In fact, recent studies have cited ketamine as a potential therapy for depression, including treatment resistant varieties, as well as alcohol use disorders. Historical PerspectiveKetamine was first discovered by Parke-Davis laboratories in 1962 as part of an effort to create a powerful, novel anesthetic agent with analgesic properties.3 The group initially produced phencyclidine, more commonly referred to as PCP, and while this substance did induce the desired effects, it produced dangerously severe excitation and delirium in a significant percentage of the trial population. After the failure of phencyclidine, the group continued to engineer derivatives and eventually created CI-581, a fused ketone and amine, later combined as the drug name ketamine, that produced cataleptic, analgesic and anaesthetic action with a much shorter duration than PCP, without the detrimental excitations.3 Ketamine proved to be a very powerful substitute as it produced “potent anesthesia, sedation and analgesia while maintaining cardiopulmonary stability and airway patency.”2 In the early years after ketamine was developed, significant research and clinical trials were performed. In 1963, ketamine was patented as a veterinary anesthetic. Following this application, ketamine was later introduced as an FDA-approved prescription medication under the brand name Ketalar in 1970 and was widely used as an anesthetic throughout the Vietnam war.2,3 By the end of the war, ketamine was regularly abused and associated with the psychedelic experiences sought by most recreational users. However, beginning in the 1990s, researchers began to rediscover ketamine as a potential source of psychopharmacological treatments.Pharmacological Profile of KetamineKetamine is a general anesthetic used for short term diagnostic procedures and surgery.4 However, expanded use in medical settings has been limited due to its negative psychological effects, including vivid hallucinations, agitation, and confusion.4 Ketamine’s mechanism of action is different from most hypnotic, antidepressant and mood disorder treating pharmaceuticals such as benzodiazepines, as ketamine does not interact with GABA receptors. Its specific and detailed physiological impacts have not been entirely explored, but at a basic level, ketamine is a noncompetitive (allosteric) antagonist of N-methyl-D-aspartate (NMDA) receptors.2 There are several theories as to its true mechanism of effect after binding, but none have been supported by significant empirical data. One theory on the effects of ketamine explores its relationship to glutamate, a major amino acid neurotransmitter that is often linked with many mood and anxiety disorders.5 Glutamate is thought to cause changes in mood and psychological state through a network of signaling involving the binding of glutamate to NMDA receptors. As ketamine is able to allosterically inhibit the NMDA receptors, it has been proposed that ketamine prevents the binding of glutamate, often producing antidepressant and other neuromodulation effects. Another more recent theory proposed that ketamine is involved in rewiring brain circuitry and changing synaptic growth to influence mood and recovery from depression.6 There are two main types of ketamine that have been used to produce various clinical effects. Racemic ketamine, often infused directly into the bloodstream, is a blend of “S” and “R” mirror ketamine molecules.7 Esketamine, often given as a nasal spray, only contains the “S” ketamine molecule and was approved by the FDA this past March under the name “Spravato.”7 S-ketamine appears to be a better long term choice for treating clinical disorders as it appears to be less prone to causing psychomimetic side effects, such as derealization and hallucinations.8 However, significantly more research must be done to confirm this finding. Ketamine’s systematic effects are quite interesting. At low and anesthetic doses, ketamine produces increased blood pressure and heart rate and does not cause any significant respiratory depression, preserving the upper airway reflexes.3 However, ketamine can induce several negative side effects in certain populations. These overall effects have been recorded by various researchers and can be organized into four main categories: (1) intoxication, similar to other sedatives and anesthesia agents; (2) perceptual changes and disturbances in auditory, visual, and somatosensory domains as well as feelings of dissociation, depersonalization and derealization; (3) delusions, including misinterpretation and thought disorders; and (4) other general negative symptoms such as an inability to speak.3 Another key characteristic of ketamine is its fast absorption time. Ketamine has a maximum plasma absorption range from three to thirty minutes, depending on the mode of delivery.3 In addition to fast uptake time, ketamine binds strongly to receptors, meaning that ketamine exerts its influence very quickly upon administration. Solution to Treatment Resistant Depression?Depression is a sometimes episodic and often chronic mental illness that can feature discrete symptomatic periods separated by periods of apparent wellness.8 It is the third leading cause of disability worldwide affecting close to 350 million individuals.9,10 Depression in individuals can have varied causation and may be treated differently for different people. Because of this non-uniformity of causation and manifestation of depression, it is imperative that these underlying factors be examined to ensure that all relevant issues are taken into account before treatment. Major depressive disorder (MDD), a significant diagnostic category of depression, is often associated with frequent relapses and incomplete recovery and remission.11 Quite frequently, individuals suffering from MDD are treated with psychopharmacological agents widely available such selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), and other antipsychotic medicals.12 Unfortunately, the majority of these agents take weeks to take effect and in 10-20% of patients, these antidepressant medications do not result in an improvement in symptoms. This untreatable population falls under the umbrella of treatment resistance. While traditional treatment has not proven to be effective in these cases, research on less traditional treatments may provide alternative treatment options in these cases. In recent years, multiple studies have linked ketamine to antidepressant effects, particularly in cases of treatment resistant depression. In a study by Murrough et al in 2013, researchers observed an 89% improvement in depression questionnaires 24 hours post ketamine administration in a population of adults with treatment resistant depression.5 Furthermore, in a study conducted by Segmiller et al, three of six participants suffering from treatment resistant depression that were treated using ketamine showed both short term and long term improvements in his/her outlook and symptoms.13 In addition to its prospective ability to alleviate some symptoms of treatment resistant depression, ketamine’s rapid absorption and effect can induce a response in several hours, providing a rapid, novel treatment compared with most antidepressant regimens, which take weeks for noticeable effect. While several of these studies examine a very small sample size, early results suggest promise for future treatment and research. While the majority of depression-related ketamine research has examined its effects, there has been very little work done to examine the underlying mechanism behind the effects. In an effort to better understand this relationship, in April 2019, Conor Liston at Weill Cornell Medicine examined synaptic changes in depressed mice that resulted from ketamine administration. Liston stressed mice in two distinctly different ways, observed the results of the stress, and then administered ketamine. He found that the ketamine seemed to partially reverse the effects of stress, regrowing synapses where stress had destroyed them. Improved behavior and circuit function were observed in the mice within a three hour span post administration, and synapses began to regrow between twelve and twenty four hours.14 While synapses regrow rapidly, they often deteriorate if not maintained properly. Liston himself admits that while the findings are very promising, there is still a great deal of additional study needed. Liston states, “Our findings open up new avenues for research…that could include drugs that stabilize the synapses or behavioral therapies that engage the healed synapses and circuits, thereby strengthening them.”14 While Liston’s study is certainly not the first study to link ketamine to antidepressant actions, it is one of the first to provide solid evidence for the mechanism that causes the effect. One slight drawback of the study is that Liston’s team only examined the effects of a single dose of ketamine and did not address how multiple doses would affect an individual. It is unclear whether multiple doses would result in an increased rejuvenation of synapses or cause some sort of regression. Even considering its limitations, the study provides empirical supporting evidence and a solid jumping off point for future clinically directed research. Treatment of AlcoholismSubstance overuse and abuse disorders such as alcohol use disorder and other substance use disorders are among the leading causes of avoidable mortality and morbidity worldwide, affecting 19.7 million individuals in the United states over the age of 12.15,16 Additionally, 74% of those who suffer from a substance abuse disorder are simultaneously suffering from an alcohol use disorder.16 As a result, when treating for alcoholism, researchers must ensure that they examine the whole clinical formulation, treating all aspects and angles to ensure nothing is missed. A treatment serving as a suppressor of desire to consume alcohol in an individual may prove to be an ineffective therapy for the comorbid condition. A study published in November 2019 suggests that ketamine could serve as a disruptor of negative alcohol consumption patterns in adults. The study focused on addiction as a memory disorder that relies on maladaptive reward memories (MRMs) to develop and maintain these addictions.17 MRMs are learned associations that are created between drug predictive environmental stimuli such as the look, taste and smell of a substance, and the specific drug reward that the nervous system experiences.18 These memories were long thought to be unalterable and current treatments for substance abuse usually focus solely on learning adaptive behavior to suppress the desires and cravings. However, recent research has examined memory reconsolidation, a process involving the reactivation and destabilization of old, long-term memories to update themselves through the incorporation of new information19. Their findings suggest that through memory consolidation it is possible to selectively target and weaken the memory cues, damaging the network of MRMs and weakening the response to the behavior.20 The research team hypothesized that triggering MRMs in individuals that drank dangerous amounts of alcohol and immediately administering ketamine would disrupt the retrieval of associated memories and result in a reduced desire to drink. To test this hypothesis, the group gathered 90 individuals who admitted that they drank too much alcohol, but were not formally diagnosed with an alcohol use disorder (Anderson), and split them into three groups: (1) induced retrieval and administered ketamine (RET+KET group); (2) no retrieval and administered ketamine; and (3) retrieval and administered placebo (saline). The results were quite intriguing. In the RET+KET, there was a significant reduction in desire to drink beer placed in front of them post ketamine administration vs. pre ketamine administration. There was no significant change noted in the control group.19 The RET+KET group also showed a significant drop in the number of binges post manipulation while the control group showed no difference. Additionally, to ensure that the outcomes examined were not simply short term, the research team continued to follow subjects up to nine months after the manipulation. Researchers observed reductions in weekly alcohol drinking in all groups, with no evidence of rebound.20 This cross-group trend was likely due to some sort of self-awareness and initiative following their admittance to dangerous alcohol use. Most significantly, the RET+KET group had cut their average alcohol consumption in half by the nine month check in. This data suggests a notable, potential relationship between ketamine administration and reduction of alcohol consumption and the results aligned with a therapeutic mechanism grounded in reconsolidation interference.20 Future DirectionsKetamine holds a great deal of promise as a potential source of psychological treatments due to its rapid absorption and effect timing, as well as potential long term results. Unlike other antidepressants or MRM reducing treatments that can take weeks to yield results, individuals who received ketamine therapy began to show results hours after administration for both antidepressant action and reduction in alcohol consumption. These findings suggest many possible avenues of exploration for future effective and efficient treatments to psychological disorders. As substance and alcohol use disorders are very often comorbid with depression and other mood disorders, treatment of both conditions simultaneously could be quite advantageous, and alleviation of one condition with ketamine treatment may lead to improvements in the other. While the future does seem bright for clinical use of ketamine, there continue to be impediments to widespread acceptance due to ketamine’s stigma, as it is an addictive narcotic that continues to be abused as a recreational drug. Clinical trials and significant research on substances such as ketamine can be expensive, often requiring the support of grant money. It is often much more difficult to get this limited funding for a controversial pharmaceutical like ketamine compared to treatments without negative associations. In addition, there are several aspects of ketamine treatment that require a great deal more research before any real conclusion can be made about its true efficacy. It is clear that there is still significant work to be done before ketamine could be a widely accepted psychological treatment. Based on the evidence provided by recent studies, the use of ketamine as a potential therapy for depression, including treatment resistant varieties, as well as alcohol use disorders, has the potential to significantly benefit individuals affected by these conditions as an alternative to traditional treatments. Future clinically directed research is warranted to determine whether ketamine treatment is the psychological breakthrough that will best address these conditions. References1.) U. (n.d.). Drug scheduling. Retrieved February 24, 2020, from https://www.dea.gov/drug-scheduling 2.) Li, L., & Vlisides, P. E. (2016). Ketamine: 50 Years of Modulating the Mind. Frontiers in human neuroscience, 10, 612. https://doi.org/10.3389/fnhum.2016.00612 3.) Mion, G. (2017). History of anaesthesia: The ketamine story – past, present and future. European Journal of Anesthesiology, 34(9), 571-575. doi:10.1097/EJA.0000000000000638 4.) N. (n.d.). Ketamine. Retrieved February 24, 2020, from https://pubchem.ncbi.nlm.nih.gov/compound/Ketamine 5.) Murrough, J. W., Perez, A. M., Pillemer, S., Stern, J., Parides, M. K., aan het Rot, M., Collins, K. A., Mathew, S. J., Charney, D. S., & Iosifescu, D. V. (2013). Rapid and longer-term antidepressant effects of repeated ketamine infusions in treatment-resistant major depression. Biological psychiatry, 74(4), 250–256. https://doi.org/10.1016/j.biopsych.2012.06.022 6.) Moda-Sava, R. N. (2019). Sustained rescue of prefrontal circuit dysfunction by antidepressant-induced spine formation. Science, 364(6436). doi:10.1126/science.aat8078 7.) Meisner, R. C. (2019, May 22). Ketamine for major depression: New tool, new questions [Web log post]. Retrieved February 24, 2020, from https://www.health.harvard.edu/blog/ketamine-for-major-depression-new-tool-new-questions-2019052216673 8.) Paul, R., Schaaff, N., F. P., Moller, H., & T. F. (2009). Comparison of racemic ketamine and S-ketamine in treatment-resistant major depression: Report of two cases. The World Journal of Biological Psychiatry, 10(3), 241-244. doi:https://doi.org/10.1080/15622970701714370 9.) Corriger, A., & Pickering, G. (2019). Ketamine and depression: a narrative review. Drug design, development and therapy, 13, 3051–3067. https://doi.org/10.2147/DDDT.S221437 10.) Ishak, W. W., Greenberg, J. M., & Cohen, R. M. (2013). Predicting relapse in major depressive disorder using patient-reported outcomes of depressive symptom severity, functioning, and quality of life in the Individual Burden of Illness Index for Depression (IBI-D). Journal of affective disorders, 151(1), 59–65. https://doi.org/10.1016/j.jad.2013.05.048 11.) Serafini, G., Howland, R. H., Rovedi, F., Girardi, P., & Amore, M. (2014). The role of ketamine in treatment-resistant depression: a systematic review. Current neuropharmacology, 12(5), 444–461. https://doi.org/10.2174/1570159X12666140619204251 12.) M. (2018, February 03). Depression (major depressive disorder). Retrieved February 24, 2020, from https://www.mayoclinic.org/diseases-conditions/depression/diagnosis-treatment/drc-20356013 13.) Segmiller, F., Rüther, T., Linhardt, A., Padberg, F., Berger, M., Pogarell, O., Möller, H.‐J., Kohler, C. and Schüle, C. (2013), Repeated S‐ketamine Infusions in Therapy Resistant Depression: A Case Series. The Journal of Clinical Pharmacology, 53: 996-998. doi:10.1002/jcph.122 14.) Makin, S. (2019, April 12). Behind the Buzz: How Ketamine Changes the Depressed Patient’s Brain. Scientific American. 15.) Torregrossa, M. M., Corlett, P. R., & Taylor, J. R. (2011). Aberrant learning and memory in addiction. Neurobiology of learning and memory, 96(4), 609–623. https://doi.org/10.1016/j.nlm.2011.02.014 16.) Thomas, S. (2020, February 3). Addiction statistics: Drug & substance abuse statistics. Retrieved February 24, 2020, from https://americanaddictioncenters.org/rehab-guide/addiction-statistics 17.) Sanders, L. (2019). Ketamine cultivates new nerve cell connections in mice. Science News. 18.) Hyman, S. E. (2005). Addiction: A Disease of Learning and Memory. The American Journal of Psychiatry, 162(8), 1414-1422. doi:https://doi.org/10.1176/appi.ajp.162.8.1414 19.) Lee J. L. (2009). Reconsolidation: maintaining memory relevance. Trends in neurosciences, 32(8), 413–420. https://doi.org/10.1016/j.tins.2009.05.002 20.) Das, R.K., Gale, G., Walsh, K. et al. Ketamine can reduce harmful drinking by pharmacologically rewriting drinking memories. Nat Commun 10, 5187 (2019). https://doi.org/10.1038/s41467-019-13162-w Suggested Reading from Inquiries Journal
Inquiries Journal provides undergraduate and graduate students around the world a platform for the wide dissemination of academic work over a range of core disciplines. Representing the work of students from hundreds of institutions around the globe, Inquiries Journal's large database of academic articles is completely free. Learn more | Blog | Submit Latest in Health Science |