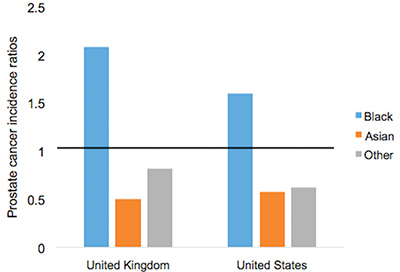
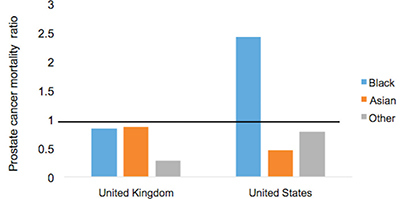
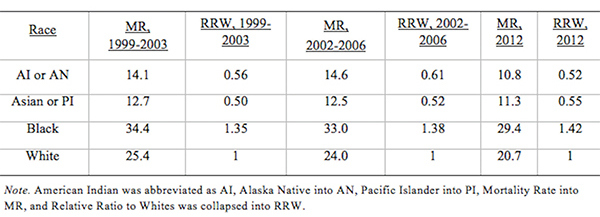
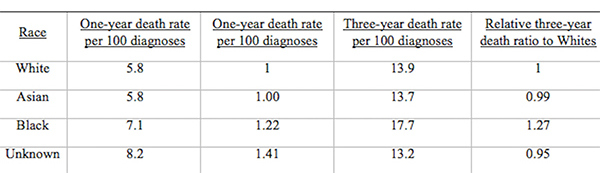
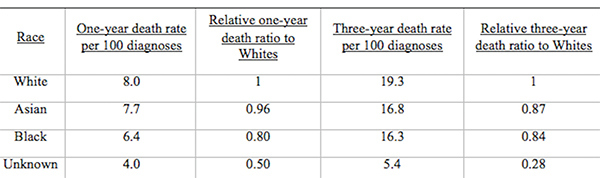
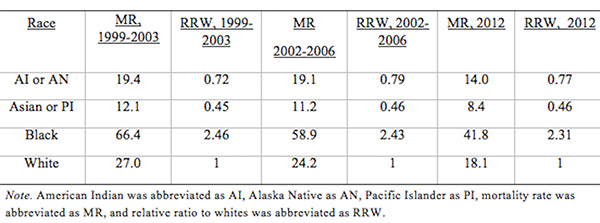
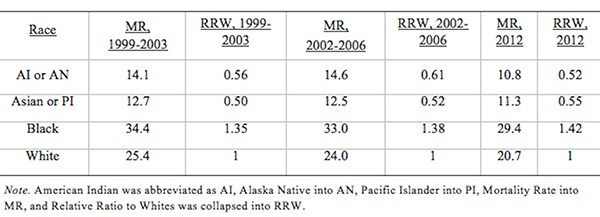
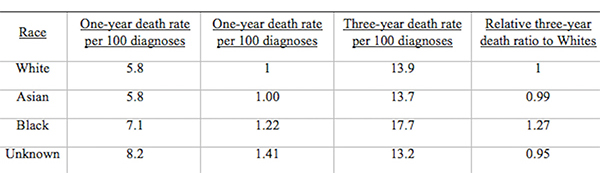
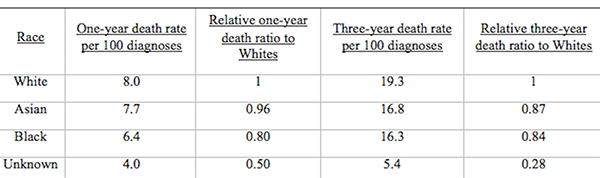
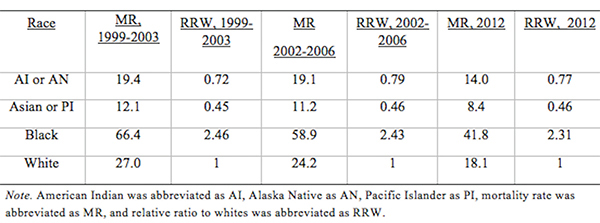
From Discussions VOL. 12 NO. 2Do Healthcare Systems Discriminate? A Comparative Policy Analysis of Health Inequality in the United States and United KingdomDiscussionTrend 1: The U.S. Achieves Lower Overall Mortality RatesThese results raise two prominent questions: Why is the U.S. better at overall breast and prostate cancer mortality than the U.K., and why is the U.S. worse at treating these conditions in Black individuals than the U.K? Three key explanations for lower overall U.S. mortality rates may explain the first trend: emphasis on early diagnosis and treatment, provision of treatments regardless of their cost-effectiveness, and emphasis on intensive intervention in all age groups. Most critical is likely the major discrepancy in screening policies. Women under 50 years of age are not offered routine breast cancer screening in the U.K., with the nation's screening program inviting women between the ages of 50 and 70 to have a mammogram every three years (NHS Breast Screening Programme, 2015). In contrast, American women between the ages of 40 and 44 are offered the choice to begin screening, with annual mammograms strongly encouraged for women aged 45 through 54, and biennial mammograms for women over the age of 55 (Oeffinger, Fontham, Etzioni, Herzig, Michaelson, et al., 2015). "As a result, the U.S. successfully diagnoses and treats prostate cancer at an earlier stage but at the cost of extensive overdiagnosis and treatment." Scientific evidence strongly suggests that the earlier a cancer is diagnosed and treated, the greater the rise in longevity and quality of life (Baron, 2009, p. 12). A commission tasked with explaining the relatively poor cancer outcomes in England therefore concluded that "delays in diagnosing cancer are a major reason why survival rates are worse in England" (Baron, 2009, p. 12). Figure 5. Age-standardized prostate cancer incidence ratios relative to White in-country reference group, which is highlighted by the black line, in the U.S. and U.K, 2002 to 2006. This trend is not exclusive to breast cancer; the discrepancies in prostate cancer also can be partially explained by early screening and diagnosis. Prostate-specific antigen (PSA) screening detects levels of an enzyme in the blood to predict the possibility of prostate cancer (American Cancer Society, 2015). However, this screening method is controversial because of its high miss (false negative) rate; 20% of men with prostate cancer have normal PSAs. Furthermore, there is an even higher rate of false positives; in order to avert just one death from prostate cancer, 27 men must be diagnosed, some of whom will possess innocuous forms of the cancer that would not have generated problems if left undetected (van Rij & Murphy, 2014). If PSA screening were introduced in the U.K., prostate cancer deaths could be reduced by 21%, yet it would lead to significant overdiagnosis and overtreatment, as 40% of cases are unlikely to progress to a fatal stage (van Rij & Murphy, 2014). Figure 6. Age-standardized prostate cancer mortality ratios relative to the in-country White reference group, which is highlighted by the black line, in the U.S. and U.K, 2002 to 2006. The NHS has therefore decided that PSA screening is not a cost-effective tool, opting not to promote the mechanism unless an individual requests it. In contrast, the American Cancer Society recommends PSA screening at age 50 for men of average risk, age 45 for men at high risk with one diagnosed relative, and age 40 for men at very high risk with more than one diagnosed relative (American Cancer Society, 2015). If the blood test returns a normal value, PSA screening is subsequently encouraged every two years; if the blood test returns a higher than normal level, patients are encouraged to pursue screening annually. As a result, the U.S. successfully diagnoses and treats prostate cancer at an earlier stage but at the cost of extensive overdiagnosis and overtreatment. These policy decisions lead to lower mortality rates but also contribute to the $280 billion in healthcare spending that is wasted annually on unnecessary care like false positives with PSA screening (Barton, 2009, p. 15). The second critical discrepancy yielding better outcomes in the U.S. is a relatively weaker focus on cost-effectiveness. In the U.K., NICE regulates available treatments in order to maximize costeffectiveness, resulting in the rejection of several drugs that only marginally extend life. One such Herceptin-style cancer drug, Kadeyla, provides six months of extra life for £90,000 ($136,670) per treatment, resulting in an estimated value of £138,000 to £153,000 ($209,560 to $232,338) per QALY (Adam, 2014). NICE therefore rejected this treatment for falling above its £30,000 ($45,556) per QALY guideline, and although patients can petition the cancer drugs fund to purchase Kadeyla, many declined the opportunity to pursue the stressful and arduous appeal process. In fact, the cancer drugs fund will be phased out by March 2016, having already removed 43 previously funded therapies over nine months in 2015 (Elvidge, 2015). An estimated 986 breast cancer patients alone will miss out on an opportunity to improve the length and quality of their lives because of these costeffectiveness decisions (Elvidge, 2015). The third factor explaining U.S. superiority in mortality rates is intensive intervention at all ages. Data from the U.K. suggest that ageism may explain some of its higher mortality rates. According to a parliamentary commission, nearly 15,000 people over the age of 75 die prematurely from cancer because they are less likely to be screened, diagnosed, and intensively treated (Baron, 2009, p. 15). Cancer patients aged 55 to 64 are 20% more likely to survive for at least one year following diagnosis than their 75+ counterparts, and researchers concluded that this discrepancy is not entirely explained by elevated co-morbidity and frailty (Baron, 2009, p. 10). Instead, older individuals are less likely to be offered intensive cancer treatments, increasing their mortality risk. The fact that the country does not even measure mortality rates for patients above the age of 75 indicates that the health system is focused on diagnosing and treating cancer in younger populations. This often comes at the expense of the older population, which may have many years of quality life remaining after treatment. There are still other factors inherent in the health care system that may explain the poor cancer outcomes for the U.K. relative to those of the U.S. The infamous "postcode lottery" of the NHS suggests that access to treatment depends partially upon the CCG in which an individual resides. Some CCGs are more effective at contracting with providers to control costs and maximize quality, which manifests in better outcomes for the population. The proportion of individuals dying from cancer is 61% higher in the worst-performing CCG than in the best-performing CCG, providing evidence of the disparities in socioeconomic status and access to quality health care across regions of the U.K. (McDonnell, 2015). Delays in diagnosis and treatment plague the NHS, which set a target of 85% of individuals treated within 62 days of a GP referral. However, the 83.1% average for the bottom quintile of CCGs is significantly lower than the 86% average for the top quintile. In recent years, the NHS has greatly improved these rates, but these improvements are not expected to compensate for the deficits in screening programs and available treatments (McDonnell, 2015). Trend 2: The U.S. Experiences Greater Inequality of OutcomesIn both countries, Black women have higher rates of breast cancer mortality than White women. While some researchers have proposed that Black women are predisposed to certain manifestations of breast cancer through genetics, current data are insufficient to warrant decisive conclusions (American Cancer Society, 2015). This trend may instead be explained by a number of potential factors. Since younger Black women under the age of 45 have a greater incidence of breast cancer than younger White women, late stage cancers that respond less favorably to treatment are expected to develop at higher rates in Black women in the U.K., which does not screen before the age of 50 (American Cancer Society, 2015). This explanation does not exist for the U.S., which begins screenings at age 40 for women. In both countries, however, Black women tend to occupy a lower socioeconomic position, predisposing them to greater rates of breast cancer through poorer diet and exercise habits, environmental factors, and a lack of health education. Limited financial access to diagnosis and treatment may be another barrier, exclusive to the U.S, as a result of the relatively lower socioeconomic position. Despite the higher mortality rates for Black citizens of both countries, it is clear that Black women in the U.S. face relative mortality ratios that are greater than those faced by Black women in the U.K. Furthermore, Black men have even higher relative mortality ratios for prostate cancer in the U.S. than than those of Black men in the U.K. These trends suggest that the U.S. healthcare system is worse than the NHS at successfully addressing gendered cancers in Black individuals. There are several possible explanations for the poorer U.S. outcomes, including the magnified intersectional effects of the lower socioeconomic status of Black Americans, general mistrust of the medical system, racial discrimination, and a lack of financial access to care. Socioeconomic status certainly plays a role in determining health outcomes, as mortality rates are two to three times higher for low-income Black Americans relative to middle-income Black Americans (Williams & Jackson, 2005). The proportion of Black and Hispanic families below the poverty level is twice that of White families in the U.S., explaining some of the U.S. discrepancy as differences in socioeconomic access rather than solely racial bias (Barton, 2009, p. 30). Those in a lower socioeconomic position tend to have lower rates of education, thus reducing their familiarity with self-care and the importance of healthy lifestyles. Residential segregation continues to play a significant role in the lives of Black Americans, limiting access to healthy food, safe housing, good education, and long-term employment opportunities that improve one's financial position (Williams & Jackson, 2005). All of these factors coalesce to produce poorer health outcomes among those occupying lower socioeconomic positions. The differences in outcomes, however, extend beyond socioeconomic factors to include fundamental suspicion of the medical system, the roots of which are entrenched in American history. From 1932 to 1972, the U.S. Public Health Service experimented on 399 Black Alabama sharecroppers with late stage syphilis. Researchers conducted their study even after the discovery of penicillin, never informing participants of their diagnosis or treatment availability, in order to determine the adverse effects of syphilis on Black individuals (The Tuskegee Timeline, 2013). The lasting effect of the Tuskegee syphilis experiment is a general suspicion of the medical profession, with race identified in studies as a significant determinant of patient satisfaction. Even when controlling for income and type of plan in which an individual is enrolled, Black Americans were 19% less likely to trust their providers (Hunt, Gaba, & Lavizzo-Mourey, 2005). The perception of racial discrimination warrants attention because it influences patient willingness to seek and follow treatment regimens, ultimately shaping health outcomes. However, there is evidence that actual racial discrimination is present within the U.S. healthcare system. Black patients are less likely than their clinically similar White counterparts to receive preventative and diagnostic services for cancer (Williams & Jackson, 2005). Even when researchers adjusted for socioeconomic status, breast cancer stage, and age, Black women observe a 22% higher mortality rate (Newman, 2005). Black women undergoing adjuvant chemotherapy regimens were more likely to experience delays in their treatment schedules, resulting in a lower overall dose intensity and poorer outcomes. While 67.1% of White American women utilize mammography between the ages of 40 and 49, only 60.9% of Black American women have at least one mammogram during that time (Newman, 2005). This discrepancy is also manifested in the use of PSAs by race, with 44.4% of White American men completing a PSA while only 35.2% of Black Americans received the screening (American Cancer Society, 2015). The disparate provision of life-extending diagnoses and treatments may partially explain the differences in mortality outcomes between Black and White Americans. One potentially significant factor contributing to this disparity is that Black Americans are more likely to lack medical insurance and rely instead on Medicaid (Newman, 2005). Health insurance is one of the most critical determinants of health outcomes, and people who possess private health insurance are more likely both to receive health care and to access diagnostic and treatment tools at an earlier disease stage (Weisfeld & Perlman, 2005). For example, of men with health insurance, 43.5% had a PSA within the past year, compared to only 13.9% of men without health insurance (American Cancer Society, 2015). This trend is true regardless of race – the relative risk of death within three years for breast cancer was 19% to 44% higher in groups of uninsured patients relative to clinically similar insured patients (Weisfeld & Perlman, 2005). Inequality of outcomes can be partially explained by the fact that Black Americans compose a greater proportion of the low-income groups that lack adequate access to health insurance and, thus, medical services. Although Medicaid is provided to low-income populations, physicians can refuse to accept new Medicaid patients and limit access to healthcare services. Medicaid co-payments were initially introduced to deter excess use of healthcare services, but consumers tend to use fewer essential medical services when facing the financial barrier. Co-payments for prescription drugs reduce the amount of prescriptions filled, but they lead to a 78% increase in adverse events – including hospitalization, nursing home admission, and death – for the population, which becomes more expensive than simply providing the initial prescriptions (Ku, 2005). Co-payments for substance-abuse services led to an initial reduction in the amount of treatment sought but a long-term increase in the rates of relapse, again backfiring in terms of money saved (Barton, 2010, p. 11). In stark contrast, the U.K. provides services to residents that are free at the point of service, thereby eliminating this financial barrier to care. All Black residents of the U.K. have access to care, regardless of socioeconomic status, potentially explaining much of the discrepancy between U.S. and U.K. relative Black to White mortality ratios. "In the U.S., fundamental change in the financial structure of the healthcare system is needed." These data raise tough questions for each country. If the U.K. wants to achieve better mortality rates overall, it must aggressively screen, diagnose, and treat patients, even if those policies lead to reduced cost-effectiveness through overtreatment. It must also raise the financial threshold per QALY that currently governs NICE's cost-effectiveness decisions and intervene when clinically appropriate at all patient ages. In the U.S., fundamental change in the financial structure of the healthcare system is needed. Financial access to care plays a significant role in determining utilization of screening programs and preventative regimens that maximize quality of life and longevity. The PPACA has extended health insurance to all Americans, but the fundamental financial barriers to care – including copayments, deductibles, and coinsurance – remain as deterrents to medical care that particularly limit access for Black Americans. A system that is free at the point-of-service will most likely lead to more equitable outcomes. Historical racial discrimination and suspicion of the medical profession must be tackled through increased diversity of health professionals and improved cultural competence. While these challenges are formidable, public policy can play a powerful role in reducing health inequalities. AcknowledgementsI would like to thank Dr. Danielle Parker of Wake Forest University and Dr. Nigel F. B. Alligton of the University of Cambridge for their mentorship. The Anna Julia Cooper Center at Wake Forest University sponsored this research. ReferencesAdam, J. (2014). Final appraisal determination: Trastuzumab emtansine for treating HER2positive, unresectable locally advanced or metastatic breast cancer after treatment with trastuzumab and a taxane. National Institute for Health and Care Excellence, 1-50. American Cancer Society. American Cancer Society recommendations for prostate cancer early detection. (2015, January 6). Retrieved from http://www.cancer.org/cancer/prostatecancer/moreinformation/prostatecancerearlydetection/prostate-cancer-early-detection-acsrecommendations American Cancer Society. Cancer prevention & early detection facts & figures (Rep.). (2015). Retrieved from http://www.cancer.org/acs/groups/content/@research/documents/ webcontent/acspc-045101.pdf Baron, J. (2009). All party parliamentary group on cancer: Report of the all party parliamentary group on cancer's inquiry into inequalities in cancer (Rep.). Sponsored by We Are MacMillan Cancer Support. Retrieved from http://www.macmillan.org.uk/Documents/GetInvolved/Campaigns/APPG/ BritainAgainstCancer2009/CancerInequalitiesReport.pdf Barton, P. L. (2010). Understanding the U.S. health services system, fourth edition. Foundation of the American College of Healthcare Executives. Health Administration Press: Chicago, Illinois. Cancer Research UK. Breast cancer mortality statistics. (2014a, September 8). Retrieved from http://www.cancerresearchuk.org/healthprofessional/cancer-statistics/statistics-by-cancer-type/breast-cancer/mortality#heading-Zeroaf Cancer Research UK. National Health Service: NHS Breast Screening Programme. (2015). Retrieved from http://www.cancerscreening.nhs.uk/breastscreen/index.html Cancer Research UK. Prostate cancer mortality statistics. (2014b, September 8). Retrieved from http://www.cancerresearchuk.org/healthprofessional/cancer-statistics/statistics-by-cancer-type/prostate-cancer/mortality#heading-Zero Centers for Disease Control and Prevention. The Tuskegee timeline. (2016, February 19). Retrieved from http://www.cdc.gov/tuskegee/timeline.htm Centers for Disease Control and Prevention WONDER. (2012). U.S. cancer statistics, 1999-2012. (2016). Retrieved from http://wonder.cdc.gov/cancer.html Claxton, K., Martin, S., Soares, M., Rice, N., Spackman, E., Hinde, S., Devlin, N., Smith, P. C., et al. (2013, June 10). Methods for the estimation of the NICE cost effectiveness threshold: revised report following referees comments (Rep.). Retrieved from https://www.york.ac.uk/media/che/documents/reports/resubmitted_report.pdf Elvidge, S. (2015). NHS England cuts treatments from Cancer Drugs Fund. The Pharmaceutical Journal, 295(7879). doi: 10.1211/PJ.2015.20069298 Hunt, K., Gaba, A., & Lavizzo-Mourey, R. (2005). Racial and ethnic disparities and perceptions of health care: Does health plan type matter? Health Services Research, 40(2), 551–576. doi: 10.1111/j.1475-6773.2005.00372. Jack, R. H., Davies, E. A., & Møller, H. (2009). Breast cancer incidence, stage, treatment and survival in ethnic groups in South East England. British Journal of Cancer, 100(3), 545-550. doi: 10.1038/sj.bjc.6604852 Jack, R. H., Davies, E. A., & Møller, H. (2010). Prostate cancer incidence, stage at diagnosis, treatment and survival in ethnic groups in South-East England. British Journal of Urology International, 105(9), 1226-1230. doi: 10.1111/j.1464410X.2009.08940.x Ku, L. (2005, July 7). The Center on Budget and Policy Priorities: The Effect of Increased Cost-Sharing in Medicaid (Rep.). Retrieved from http://www.cbpp.org/research/the-effect-of-increased-cost-sharing-in-medicaid McDonnell, P. (2015). NHS England: Cancer Waiting Times, August 2015 (Rep.). Retrieved from https://www.england.nhs.uk/statistics/wp-content/uploads/sites/2/2015/10/Cancer-Waiting-Times-Press-release-August2015-Provider-based-PDF-140kB.pdf National Health Service. NHS Choices: Help with health costs. (2015, October 29). Retrieved from http://www.nhs.uk/NHSEngland/Healthcosts/Pages/ universal-credit.aspx Newman, L. A. (2005). Breast cancer in African-American women. The Oncologist: Breast Cancer, 10(1), 1-14. doi: 10.1634/theoncologist.10-1-1 Oeffinger, K. C., Fontham, E. T. H., Etzioni, R., Herzig, A., Michaelson, J. S., Shih, Y. C. Walter, L. C., Church, T. R., et al. (2015). Breast cancer screening for women at average risk. The Journal of the American Medical Association, 314(15), 1599-614. doi: 10.1001/jama.2015.12783 Squires, D. A. (2012). Explaining high health care spending in the U.S.: An international comparison of supply, utilization, prices, and quality. Issue Brief: Commonwealth Fund 10, 1-14. Taylor, F., Gauher, S., Johnson, L., Brock, R., & Wittenberg, M. (2014). Understanding the New NHS. (Rep.) NHS England. BMJ Publishing Group. Retrieved from https://www.england.nhs.uk/wp-content/uploads/2014/06/simple-nhs-guide.pdf Thomson, C. & Forman, D. (2009, June). Cancer incidence and survival by major ethnic group, England, 2002-2006 (Rep.). Retrieved from http://www.cancerresearchuk.org/ van Rij, S. & Murphy, D. G. (2014). Words of wisdom. Re: Screening and prostate cancer mortality: Results of the European randomised study of screening for prostate cancer (ERSPC) at 13 years of follow-up. European Urology, 66(6), 11871188. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/25587592 Weisfeld, A. & Perlman, R. L. (2005). Disparities and discrimination in health care: An introduction. Perspectives in Biology and Medicine, 48(1), S1-S9. doi: 10.1353/pbm.2005.0046 Williams, D. & Jackson, P. B. (2005) Social sources of racial disparities in health. Health Affairs, 24(2), 325-334. doi: 10.1377/hlthaff.24.2.325 AppendixTable 1: U.S. Age-Standardized Mortality Rates for Female Breast Cancer per 100,000 People by Race and Year Table 2: U.K. Age Standardized Female Breast Cancer Mortality Rates per 100,000 People and Relative Ratios, 2002 and 2006 Table 3: Age-Standardized Male Prostate Cancer Mortality Rates per 100,000 People and Relative Ratios to Whites for 2002-2006 Suggested Reading from Inquiries Journal
Inquiries Journal provides undergraduate and graduate students around the world a platform for the wide dissemination of academic work over a range of core disciplines. Representing the work of students from hundreds of institutions around the globe, Inquiries Journal's large database of academic articles is completely free. Learn more | Blog | Submit Latest in Health Science |