Featured Article:Melatonin and Sleep Disorders: The Neurobiology of Sleep, Circadian Rhythm Sleep Disorders, and Various Treatment Methods
By
2013, Vol. 5 No. 07 | pg. 1/3 | »
IN THIS ARTICLE
KEYWORDS
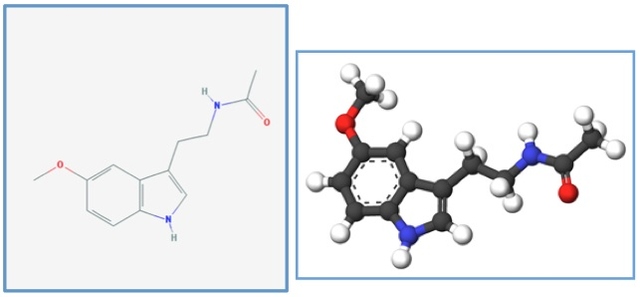
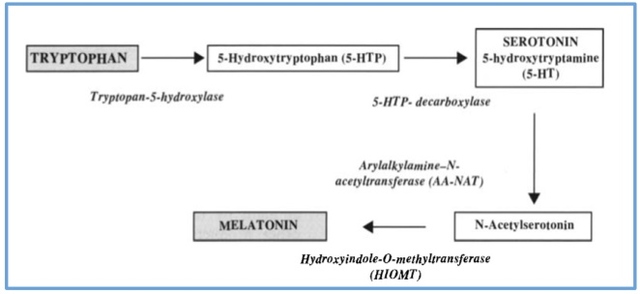
AbstractMelatonin is a lipophilic, indolic-structured compound found ubiquitously in nature. This ubiquity suggests that such an evolutionarily conserved molecule participates in phylogenetically primordial signalling mechanisms of key importance to life. Melatonin’s versatility is shown in its various physiological functions including immunomodulation, antioxidation and its role as an oncostatic agent. Synthesis mainly occurs in the pineal gland; extrapineal sites include the retina, the brain and the gut. Endogenous secretion occurs at night with a marked circadian rhythm regulated by the body’s intrinsic biological clock. This central circadian pacemaker is situated in the suprachiasmatic nucleus (SCN) of the anterior hypothalamus. Such a close association between sleep propensity and melatonin levels has led to the acceptance of this hormone as a chronobiotic and an internal ‘zeitgeber’ since it synchronizes the SCN. The sleep-wake cycle appears to be influenced by melatonin with disruption of the ascending arousal system in the hypothalamus. Sleep disorders related to disturbances in the circadian pacemaker are called circadian rhythm sleep disorders (CRSDs). They are described as a misalignment between the individual’s normal circadian rhythms and the external circadian cues such as the day/night cycle. CRSDs can be classified into two subsets: intrinsic and extrinsic. The effects of such sleep disorders may be manipulated by administering melatonin which can promote sleep and induce phase shifts. Pharmacologic treatment however needs to be focused on melatonin agonists since direct melatonin administration is not as effective due to the hormone’s short half-life. Ramelteon, agomelatine, tasimelteon and PRM (prolonged-release melatonin) are discussed as therapy for CRSDs. What is Melatonin?1.1 Nature, Structure and Source Melatonin was first discovered in 1958 by Aaron Lerner - an American dermatologist who managed to isolate it from the bovine pineal gland (Claustrat et al., 2005). The lipophilic, indolic-structured compound was so named because it was seen to induce contraction of amphibian melanophores as a result of melanin granule aggregation, thus acting as a skin-lightening factor (Barrenetxe et al., 2004, Claustrat et al., 2005). In humans, as in other vertebrates, melatonin production takes place primarily in the pineal gland – a major component of the epithalamus and evolutionarily derived from lower vertebrates’ visual system (Arendt, 1998).The pineal gland is a midline structure lying rostral to the small, upper protuberances of the midbrain’s corpora quadrigemina, known as the superior colliculi. After its release from the pineal gland, melatonin is released into the systemic circulation and gains access to various cellular compartments and tissues. These include the cerebrospinal fluid, saliva, the preovulatory follicle, semen as well as urine. It then reaches its target tissues and binds to the appropriate receptors. Having high lipid solubility and high water solubility, melatonin can cross cell membranes relatively easily (Claustrat et al., 2005, Zawilska et al., 2009). Other production sites such as the skin, the retina, the gut, platelets and the bone marrow have also been identified but these secondary sources of melatonin do not contribute significantly to physiologic blood levels (Zawilska et al., 2009). In fact, pinealectomy reduces detectable circulating melatonin concentrations considerably. However, since normal function of the mammalian circadian biorhythm is maintained in the absence of the pineal, and hence, privation of melatonin supply, this whole system is not believed to be essential to life (Arendt, 1998). Melatonin is reportedly present in organisms hailing from different stages of our life continuum. Bacteria, fungi, protozoa, invertebrates and even higher plants are capable of producing melatonin to a greater or lesser extent, generally with levels being too low to provide a consequential supply. The ubiquity of this highly evolutionarily conserved molecule suggests that its physiologic role is key to understanding basic life events implying that melatonin was probably one of the earliest compounds to appear on earth (Claustrat et al., 2005, Zawilska et al., 2009). 1.2 Biosynthesis and Secretion Melatonin (N-acetyl-5-methoxytryptamine), shown in Figure 1.1, is synthesized in the pinealocytes of the pineal gland during the night-time and has therefore been dubbed the ‘darkness hormone’ (Zawilska et al., 2009). The quantity and duration of its secretion is in fact proportionate to the length of the dark phase. Maximum amplitude of plasma levels occur at around 03.00 – 04.00 am (Claustrat et al., 2005). Since the pineal gland in mammals lost its photosensitivity through evolution, information about light cues in the environment cannot be conveyed by the usual retinal processing system. In fact, light signals are transferred to the suprachiasmatic nucleus in the anterior hypothalamus – the site of the internal biological master clock, also known as the central circadian pacemaker. This transfer of information is mediated by the retinohypothalamic tract. Such light signal parameters (including wavelength, duration and intensity) are then relayed from the SCN to the pineal gland via a number of tracts in-between (Pandi-Perumal et al., 2008, Zawilska et al., 2009). Initiation of melatonin synthesis occurs when noradrenaline is released from a peripheral sympathetic tract originating in the superior cervical ganglion and innervating the pineal gland. Postganglionic sympathetic fibres release noradrenaline which binds to adrenergic β1 receptors. Adenylate cyclase is then activated, causing intracellular cyclic AMP levels to rise and the enzyme serotonin N-acetyl transferase is then synthesized de novo (Claustrat et al., 2002). The biosynthetic pathway for the hormone starts with the amino acid precursor L-tryptophan, which is made available through the diet and taken up from the circulation into the pineal gland. Figure 2.2 shows the sequence of steps required for melatonin synthesis. Tryptophan is first converted to 5-hydroxytryptophan by the mitochondrial enzyme tryptophan hydroxylase which requires tetrahydrobiopterin coenzyme for proper catalytic activity. 5-HT is then transformed into 5-hydroxytryptamine, also known as serotonin, by the enzymatic action of 5-hydroxytryptophan decarboxylase. Melatonin formation eventually occurs via a two-step process involving N-acetylation and O-methylation by NAT and hydroxyindole-O-methyl transferase respectively. NAT is the rate-limiting enzyme for melatonin synthesis and it is precisely its activity which increases from 30-fold to about 70-fold at night. Nutritional availability of additional factors such as folate and vitamin B6, which acts as coenzyme in the decarboxylation of tryptophan, also affects melatonin production (Fournier et al., 2002; Munoz-Hoyos et al., 1996; Luboshitzky et al., 2002; Zimmermann et al., 1993). Figure 1.1. Chemical Structure of Melatonin
Figure 1.2: Schematic representation of melatonin biosynthesis
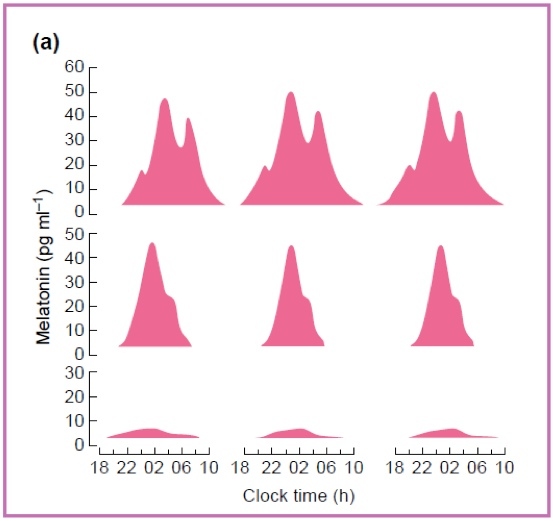
Figure 1.3: Melatonin Secretion Profile – interindividual variation present in both amplitude and secretion pattern; maximal secretion takes place at night.
1.3 Metabolism Melatonin is catabolized primarily in the liver, where it is first hydroxylated at the C6 position by a cytochrome-P450, notably the CYP1A2 isoform. It is then either sulfated, or, to a much lesser extent, glucoronated (Facciola et al., 2001; Pandi-Perumal et al., 2006). The formed conjugates are subsequently excreted in urine. This deactivation occurs rapidly, with half-life in humans varying between 10 to 60 minutes, after exogenous administration (Fourtillan, et al., 2000). Melatonin in the brain is degraded in a different manner, by oxidative splitting of the pyrrole-ring (Zawilska et al., 2009). Retinal melatonin is also metabolized differently, with deacetylation, deamination and further oxidation or reduction to 5-methoxyindoleacetic acid or 5-methoxytryptophal respectively (Grace et al., 1991). 1.4 Physiological Functions Melatonin, being a versatile biological signal, has various physiological functions among which are its antioxidant properties and its immunomodulatory role. Melatonin is capable of scavenging toxic hydroxyl radicals and is also indirectly involved in the upregulation of antioxidative enzymes (such as superoxide dismutase and glutathione peroxidase) and the downregulation of pro-oxidant enzymes (including nitric oxide synthases and 12-lipo-oxygenases). Enhanced production of certain interleukins and the fact that lymphoid cells may harbor melatonin-binding sites contribute evidentially to melatonin’s role in immunity. Its antiproliferative actions, demonstrated through several experimental studies, have also established it as a valid oncostatic agent. (Pandi-Perumal et al.,2006). Melatonin is also implicated in cardiovascular regulation and depression. In the former, it is known to have a hypotensive action and low serum levels are detected in both cases. Sexual maturation and reproduction, specifically pubertal inhibition, as well as obesity and energy expenditure are also affected (Claustrat et al., 2005; Pandi-Perumal et al., 2006). What is perhaps most striking about melatonin, however, is that it is a chronobiotic – a hormone that is capable of influencing the biological clock, also known as the central circadian pacemaker (Arendt et al., 2008). This important site in the body is located in the anterior hypothalamus, more specifically in the SCN. An endogenous, genetically-encoded rhythmicity is generated by this pacemaker, which then needs to be entrained (synchronized) by external cues, such as the light/dark cycle and meal timing, to the daily 24-hour cycle. If such ‘zeitgebers’ did not exist, circadian patterns would have their own timing and would not be precisely 24 hours long (Pandi-Perumal et al., 2008). Several studies have demonstrated evidence that has confirmed melatonin as a chronobiotic. In one of the more recent studies, a 1.5mg melatonin preparation was administered daily to eight healthy, non-smoking, male volunteers for two, 14-day trials. The circadian rhythm of sleep shifted without any change in overall sleep duration (Rajaratnam et al., 2004). 1.5 Melatonin Receptors Two classes of melatonin membrane receptors – MT1 and MT2 – were originally classified, both belonging to the G-protein coupled receptor superfamily and showing 55% amino acid homology. The MT3 subtype, more recently identified, is not a G-protein coupled receptor but the enzyme quinone reductase 2 (Barrenetxe et al., 2004; Zawilska et al., 2009). Such reductases help to prevent oxidative stress (Foster et al., 2000). While MT1 receptors seem to enable vasoconstriction, MT2 receptors have the opposite effect and cause vasodilation. Also, the firing rate of neurons is decreased when MT1 receptors are activated. The MT2 receptors, on the other hand, control phase shifts. SCN receptors are responsible for the chronobiological effects while those expressed in more peripheral regions (such as skin, liver and kidney tissues) are thought to influence vasomotor regulation and immunomodulation (Dubocovich et al., 2005; Pandi-Perumal et al., 2006). In mammals, melatonin receptor distribution is limited and expression is highest in an area of the anterior pituitary known as the pars tuberalis (Zawilska et al., 2009). Nuclear melatonin receptors have also been identified (Carlberg and Weisenberg, 1995). Correlations between sleep disorders and receptor polymorphisms in humans may exist (Claustrat et al., 2005).Continued on Next Page » Suggested Reading from Inquiries Journal
Inquiries Journal provides undergraduate and graduate students around the world a platform for the wide dissemination of academic work over a range of core disciplines. Representing the work of students from hundreds of institutions around the globe, Inquiries Journal's large database of academic articles is completely free. Learn more | Blog | Submit Latest in Health Science |