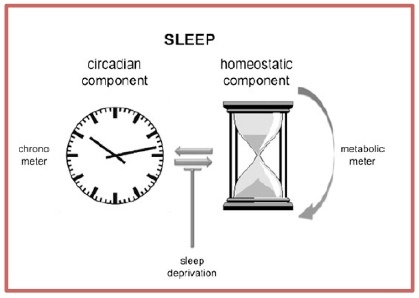
Featured Article:Melatonin and Sleep Disorders: The Neurobiology of Sleep, Circadian Rhythm Sleep Disorders, and Various Treatment MethodsNeurobiology of Sleep2.1 Introduction to Sleep Sleep is thought to be an essential, cyclic, physiologic state defined by a marked decrease in consciousness, a reduction in muscle movement and a general slowing-down of metabolism (Zisapel, N., 2010). About one-third of our lives is spent sleeping. The sequence of stages in sleep is controlled by a group of nuclei in the brainstem and various functions are attributed to this highly conserved behavior even though its exact purpose has not yet been established. Hypothetical functions include the replenishment of glycogen levels in the brain, energy conservation as body temperature falls during the night-time and more recently, consolidation of memories by increasing connection strength between synapses. (Purves et al., Neuroscience – 4th ed. 2008) Sleep in mammals occurs in two forms: rapid eye movement (REM) and non-REM (NREM), which alternate in four or five cycles lasting 70-90 minutes each (Pandi-Perumal et al., 2008). The two are distinguished by the use of polysomnography: the combination of electroencephalography (EEG), electromyography (EMG) and electroculography (EOG) measurements (Pace-Shott and Allan Hobson, 2002).2.2 The Sleep-Wake Cycle The waking state is largely controlled by what is known as the ascending reticular activating system, located in the diencephalon and the upper brainstem. One division of this ascending arousal system projects to thalamic nuclei whereas a second division extends to the lateral hypothalamus all the way to the cerebral cortex. The sleep state, in contrast, is promoted by what is known as the ventrolateral preoptic nucleus (VLPO). During sleep, its neurons become active whilst its efferents silence the ascending reticular activating system via the inhibitory neurotransmitters GABA and galanin. Afferents from the arousal system, in turn, project to the VLPO and inhibit it during wakefulness. Essentially, a feedback loop between the arousal and VLPO systems, represented by the ‘flip-flop’ switch model, exists to create dynamic state stability (Schwartz and Roth, 2008). 2.3 Homeostatic and Circadian Regulation of Sleep Sleep is controlled by two different regulatory processes: the ‘C’ circadian system, in charge of sleep induction and arousal, and the ‘S’ sleep debt homeostatic mechanism. The latter implies that in the case of sleep deprivation, a number of hours of recovery sleep need to ensue which are proportional to the loss of sleep. One substance thought to act as a somnogen (sleep-promoter) in the homeostatic regulation of sleep is adenosine. This accumulates as ATP levels are depleted and ATP is itself degraded. Both ‘S’ and ‘C’ processes interact with the SCN in the lateral region of the hypothalamus, that functions as the ‘master clock’ of the brain (Pandi-Perumal et al., 2008). The circadian regulation of sleep involves a three-stage sequence of steps starting from the SCN, which mainly projects to the nearby subparaventricular zone. This, in turn, relays information to the dorsomedial nucleus of the hypothalamus. The DMH is one of the GABAergic inputs to the VLPO and the orexin neurons (component of the ascending arousal system) located within the hypothalamus (Pace-Schott and Allan Hobson, 2002; Saper et al., 2005). Such a complex pathway provides us with the capacity to flexibly adapt physiological cycles in view of external, environmental indicators (Schwartz and Roth, 2008). The SCN, under physiologic or normal circumstances, is reset diurnally by different mechanisms, depending on the light-dark cycle. During the day it is synchronized via light signals from the retina, specifically by the circadian photopigment melanopsin. It is melatonin secretion that synchronizes the SCN at night. With loss or damage of the SCN and no external timing cues, the circadian rhythms of a range of physiological bodily processes become disrupted (Pace-Schott and Allan Hobson, 2002; Saper et al., 2005). Figure 2.1. The Circadian and Homeostatic Components of Sleep. Sleep deprivation may cause the two intricately-linked components to become uncoupled.
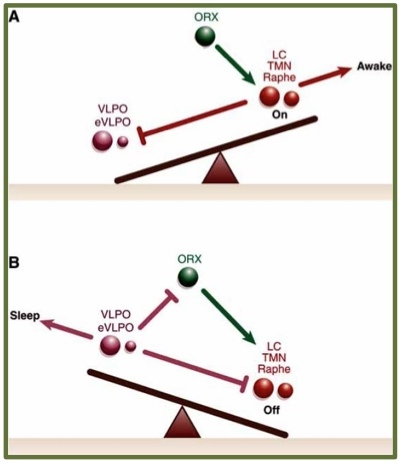
Figure 2.2: The ‘flip-flop’ switch model. A) during wakefulness: ascending arousal system activated; B) during sleep: ascending arousal system inhibited. ORX: orexin neurons; LC: locus coeruleus; VLPO: ventrolateral preoptic nucleus; eVLPO: extended VLPO; TMN: tuberomamillary nucleus.
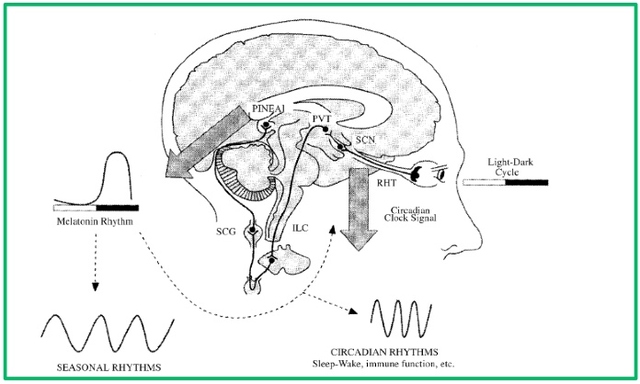
2.4 Melatonin’s role in the regulation of the Sleep-Wake cycle Sleep periodicity is adjusted to a circadian rhythm which is entrained by external cues. However, the body’s ‘internal clock’, as previously mentioned, still operates even when such ‘zeitgebers’(from the German term ‘time givers’) are removed and no information indicating the time of day is communicated. In this case, it is said to run freely and the daily 24-hour cycle lengthens to about 26. The sleep-wake cycle therefore, needs to be photoentrained to the day-night cycle so that sleep and wakefulness states are maintained appropriately. Changes in light levels must be detected by specialized photoreceptors which project to that part of the anterior hypothalamus known as the SCN via the retino-hypothalamic tract (Purves et al., Neuroscience – 4th ed. 2008; Sharma and Feinsilver, 2009). In the absence of daylight, melatonin resets the SCN during the dark cycle. It affects circadian rhythms by altering the SCN’s metabolic and electrical activity via the protein-kinase C second-messenger system (Pace-Shott and Allan Hobson, 2002; Pandi-Perumal et al., 2006). Light, however, can repress secretion of nocturnal melatonin as well as contribute to chronodisruption. The extent of this depends on duration and intensity. Maximum suppression occurs at intensities of 2000-2500 lux for 2 hours between 2:00 and 4:00am (Claustrat et al., 2005). Phases of free-running melatonin rhythms may be reset with light. Such phase shifts can be displayed as PRCs – Phase Response Curves. Melatonin and light PRCs mirror each other thereby reinforcing melatonin’s label as the ‘darkness hormone’ and thus a transmitter of time-of-day information (Arendt, 1998). It has been shown through human studies that melatonin administration, in both physiologic as well as pharmacologic doses, induces phase shifts and promotes sleep induction and maintenance (Arendt, 1998, Zhdanova et al., 1995&1996). As a rule, increased neuronal activity in the SCN and sleep proclivity result after the endogenous rise in nocturnal melatonin secretion. Synthesis and secretion of melatonin runs parallel to sleep rhythm. This association between melatonin and sleep has been confirmed in studies on blind people, whose SCN cannot be synchronized to the circadian rhythm (Lockley et al., 1997). Dim light melatonin onset (50 lux) timing is used as a consistent marker of circadian phase, even though this may vary between individuals (Cajochen et al., 2003; Sletten et al., 2010). Ultimately, melatonin is thought to regulate the sleep-wake cycle by inhibiting the brain network involved in the ascending arousal system of the hypothalamus (Shochat et al., 1998). Conditions such as aging, diabetic neuropathy, Alzheimer’s Disease as well as particular drugs such as β-blockers and NSAIDs stop synthesis of nocturnal melatonin and thus lead to impaired sleep (Zisapel, 2000). Fatigue and somnolence were also induced with daytime melatonin administration, at a time when endogenous levels in the body are minimal (Cajochen et al., 2003). Figure 2.3: Regulation of melatonin secretion – light signals are conveyed to the SCN via the retino-hypothalamic tract. At the SCN it entrains the circadian pace-maker to 24 hours and efferent circadian signals are subsequently relayed to different parts of the brain. This includes the pineal gland which is the site of melatonin synthesis. Suggested Reading from Inquiries Journal
Inquiries Journal provides undergraduate and graduate students around the world a platform for the wide dissemination of academic work over a range of core disciplines. Representing the work of students from hundreds of institutions around the globe, Inquiries Journal's large database of academic articles is completely free. Learn more | Blog | Submit Latest in Health Science |