The Epidemiology, Pathogenesis, and Treatment of Type 1 Diabetes Mellitus
By
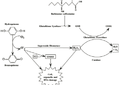
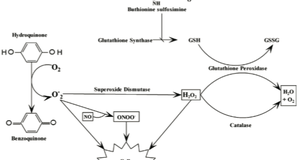
2015, Vol. 7 No. 11 | pg. 2/2 | « Treatment and PreventionPast TherapiesImmunosuppressive Therapies for Treatment of T1DMIn the mid-1980’s, Cyclosporine A was the first large-scale immunosuppressive to be studied (Grieco, Vendrame, Spagnuolo et al., 2011). Cyclosporine A reduced the activity of the immune system, decreased autoimmune responses, and lowered the incidence of T1DM (Grieco, Vendrame, Spagnuolo et al., 2011). However, renal toxicity was a common side-effect and cyclosporine A testing ceased (Stiller, Dupre, Gent et al., 1987). Present TherapiesInsulin Injections/Pumps and Glucose MonitorsCommon treatments for T1DM include frequent injections of rapid-acting insulin with meals combined with daily basal insulin (Luo, Herold & Miller, 2010) (Atkinson & Eisenbarth, 2001). Moreover, insulin pumps are widely used to treat youth with the disease (Atkinson & Eisenbarth, 2001). In the 1990’s, pediatric diabetologists were cautious when using insulin pumps for children (Maahs, West, Lawrence et al., 2010) (Luo, Herold & Miller, 2010). They observed a three-fold increase in severe hypoglycemia in subjects who were extensively treated with insulin pumps during the Diabetes Control and Complications Trial (DCCT). However, developments in our understanding of insulin’s mechanism and advancements in pump features have reduced the caution generated by hypoglycemia (Maahs, West, Lawrence et al., 2010). The development of continuous glucose monitors (CGMs) has improved glycemic outcomes in T1DM patients (Haller, Atkinson & Schatz, 2005) (Maahs, West, Lawrence et al., 2010). CGMs were evaluated in a JDRF clinical trial. The clinical trial compared T1DM adults with glycated haemoglobin (A1c) levels over 7% who were using CGMs to controls (Maahs, West, Lawrence et al., 2010). A large reduction in A1c levels was observed in adults 25 years of age or older. In contrast, A1c levels rose in controls (Maahs, West, Lawrence et al., 2010).Antigen Specific Therapies for Treatment and Prevention of T1DMAntigen specific therapies are safer than immunosuppressive therapies (Grieco, Vendrame, Spagnuolo et al., 2011). Hence, antigen specific therapies are crucial intervention techniques for T1DM patients (Achenbach, Bonifacio & Ziegler, 2005). The most thoroughly studied antigens to date include insulin and GAD65 (Gilliam, Binder, Banga et al., 2004) Antigen specific therapies use target autoantigens to eradicate pathogenic T cells and activate T regulatory cells at the target organ (Grieco, Vendrame, Spagnuolo et al., 2011). Trialnet/Diabetes Prevention Trial (DPT) conducted two studies to determine insulin’s potential in preventing the development of diabetes in T1DM patients (Grieco, Vendrame, Spagnuolo et al., 2011). Insulin was administered parenterally and orally. The study found that parenteral insulin did not decrease progression of the disease. Similarly, oral insulin did not alter overall progression of diabetes (Grieco, Vendrame, Spagnuolo et al., 2011). However, in subset of patients with high levels of insulin autoantibodies, delay in progression of T1DM was observed (Type 1 Diabetes TrialNet, 2011). Environmental Therapy for the Prevention of Islet AutoimmunityEarly exposure to foodstuffs may confer susceptibility to T1DM (Sherr, Sosenko, Skyler et al., 2008). The Trial to Reduce IDDM in the Genetically at Risk (TRIGR) documented the rates of autoantibody development and the incidence of T1DM in children, ages 10 and younger (Michels & Eisenbarth, 2011). Infants who had a first-degree relative with T1DM and possessed high-risk HLA alleles were given a highly hydrolyzed formula (treatment group) or conventional cow’s milk formula (control group) (Michels & Eisenbarth, 2011). Children who received the hydrolyzed casein-based formula had less autoantibody positivity campared to the control group (Michels & Eisenbarth, 2011), suggesting that environmental therapies may benefit T1DM patients. Future Directions for TreatmentA variety of new technologies and therapies are being developed in order to treat and prevent the development of T1DM. Novel TechnologiesIt can be argued that major advances made in T1DM care within the last quarter century have come from technology rather than biology. Technologies such as rapid glucose estimation from a drop of blood, insulin pumps and quick HbA1c results are rapidly becoming commonplace. Moreover, mathematicians, engineers, physicians, scientists and patients have combined their efforts to develop external insulin delivery devices and an "artificial pancreas." These technologies, despite the expense, have given patients freedom and autonomy when it comes to taking care of their disease themselves (Gregory et al., 2010). Stem Cell TherapiesBecause T1DM is the result of a loss of β cells in the pancreatic islets (i.e. a single cell type), it is an ideal candidate for regenerative stem cell therapy. Embryonic stem cells and induced pluripotent (iPS) stem cells are high on the list of candidate stem cells being studied to treat T1DM (Aguayo-Mazzucato & Bonner-Weir, 2010). Embryonic stem cells are pluripotent (i.e. can turn into any cell type in the body). Consequently, scientists have turned mice and human embryonic stem cells into insulin-producing β cells. Studies have shown that newly formed β cells can properly regulate blood sugar levels after being transplanted into mouse models of diabetes. However, biological hurdles (e.g. potential for formation of tumors) and ethical considerations associated with embryonic stem cells may hamper their future as therapeutic agents (Aguayo-Mazzucato & Bonner-Weir, 2010). Induced pluripotent (iPS) stem cells can be reprogrammed from normal adult skin cells and can become a multitude of cell types in the body. A 2010 study first differentiated induced pluripotent stem cells into insulin-secreting β-like cells and showed that, upon transplantation, they could properly regulate blood glucose levels (Aguayo-Mazzucato & Bonner-Weir, 2010). Genetic EngineeringGenetic engineering describes the process by which a functional gene is introduced into a new tissue or organ, allowing for the expression of a new feature or characteristic. Investigators of T1DM have evaluated the likelihood of modifying islet cells to render them resistant to immune destruction prior to transplantation. Furthermore, the possibility of altering various cell types to convert them into insulin-producing cells for later transplantation in the same individual had been investigated. However, significant research is required before genetic engineering techniques can be practically applied in the treatment of T1DM (Wong et al., 2010). ConclusionsT1DM is a chronic disease caused by autoimmune (type 1a) or spontaneous (type 1b) destruction of pancreatic beta cells, resulting in insulin deficiency. Ethnicity, genetics and environmental influences contribute to the development of T1DM.The development of T1DM comprises a complex set of events from the activation of antigen-presenting cells (APCs) that present β-cell antigens to T cells that cause islet destruction and insulin deficiency. Animal models have improved our understanding of the immunological mechanisms that underlie T1DM. Common treatments for T1DM include frequent injections of rapid-acting insulin with meals combined with daily basal insulin. Moreover, insulin pumps are widely used to treat youth with the disease. However, stronger knowledge of innate immune cell structures will aid in the discovery of a cure for T1DM in humans. ReferencesAchenbach, P., Bonifacio, E., & Ziegler, A.G. (2005). Predicting type 1 diabetes. Curr. Diab. Rep, 5, 98–103. Aguayo-Mazzucato, C., & Bonner-Weir, S. (2010). Stem cell therapy for type 1 diabetes mellitus. Nat Rev Endocrinol, 6 (3), 139-48. Akerblom, H.K., & Knip, M. (1998). Putative environmental factors in type 1 diabetes. Diabetes Metab Rev, 14, 31-67. Akira, S., Uematsu, S., & Takeuchi, O. (2006). Pathogen recognition and innate immunity. Cell, 124, 783–801. Anderson, M.S., & Bluestone, J.A. (2005). The NOD mouse: A model of immune dysregulation. Annu. Rev. Immunol., 23, 447–485. Atkinson, M.A., & Eisenbarth, G.S. (2001). Type 1 diabetes: New perspectives on disease pathogenesis and treatment. Lancet, 358, 221–229. Concannon, P., Rich, S.S., & Nepom, G.T. (2009). Genetics of type 1 A diabetes. N. Engl. J. Med, 360, 1646–1654. Dabelea, D., Mayer-Davis, E.J., & Imperatore, G. (2010). The value of national diabetes registries: SEARCH for diabetes in youth study. Curr Diab Rep, 10, 362-9. Dotta, F., Fondelli, C., & Falorni, A. (2008). Can NK cells be a therapeutic target in human type 1 diabetes? Eur J Immunol, 38, 2961–2963. Ekici, R. (2010). B cells in type 1 diabetes: Studies on cell surface antibody binding. Available at http://www.diva-portal.org/smash/get/diva2:359800/FULLTEXT01.pdf Elliott, R.B., Harris, D.P., Hill, J.P. et al. (1999). Type I (insulin-dependent) diabetes mellitus and cow milk: casein variant consumption. Diabetologia, 42 (3), 292–296. Gan, M.J., Albanese-O’Neill, A., & Haller, M.J. (2012). Type 1 diabetes: Current concepts in epidemiology, pathophysiology, clinical care, and research. Curr Probl Pediatr Adolesc Health Care, 42, 269-291. Ghazarian, L., Diana, J., Simoni, Y. et al. (2013). Prevention of acceleration of type 1 diabetes by viruses. Cell. Mol. Life Sci, 70, 239-255. Gilliam, L.M., Binder, K.A., Banga, J.P. et al. (2004). Multiplicity of the antibody response to GAD65 in Type 1 diabetes. Clin Exp Immunol, 138 (2), 337-341. Gregory, J.M. et al. (2010). Incorporating type 1 diabetes prevention into clinical practice. Clin Diabetes, 28 (2), 61–70. Grieco, F.A., Vendrame, F., Spagnuolo, I. et al. (2011). Innate immunity and the pathogenesis of type 1 diabetes. Semin Immunopathol, 33, 57-66. Haller, M.J., Atkinson, M.A., & Schatz, D. (2005). Type 1 diabetes mellitus: Etiology, presentation, and management. Pediatr Clin North Am, 52, 1553-78. Hyttinen, V., Kaprio, J., Kinnunen, L. et al. (2003). Genetic liability of type 1 diabetes and the onset age among 22,650 young Finnish twin pairs: a nationwide follow-up study. Diabetes, 52 (4), 1052–1055. Ikegami, H., Noso, S., Babaya, N. et al. (2011). Genetics and pathogenesis of type 1 diabetes: Prospects for prevention and intervention. Journal of Diabetes Investigation, 2, 415-420. Janeway, C.A., & Medzhitov, R. (2002). Innate immune recognition. Annu Rev Immunol, 20, 197–216. Kim, H.S., & Lee, M. (2009). Role of innate immunity in triggering and tuning of autoimmune diabetes. Current Molecular Medicine, 9, 30-44. Lévy-Marchal, C., Patterson, C.C., & Green, A. (2001). Geographical variation of presentation at diagnosis of type I diabetes in children: The EURODIAB study. Diabetologia, 44 (Suppl 3), B75-80. Liese, A.D., D’Agostino, R.B. Jr., Hamman, R.F. et al. (2006). The burden of diabetes mellitus among US youth: Prevalence estimates from the SEARCH for diabetes in youth study. Pediatrics, 118, 1510-8. Luo, X., Herold, K.C., & Miller, S.D. (2010). Immunotherapy of type 1 diabetes: Where are we and where should we be going? Immunity, 488-499. Maahs, D.M., West, N.A., Lawrence, J.M. et al. (2010). Chapter 1: Epidemiology of type 1 diabetes. Endocrinol Metab Clin North Am, 39 (Gan, Albanese-O’Neill & Haller, 2012), 481-497. MacKay, P., Jacobson, J., & Rabinovitch, A. (1986). Spontaneous diabetes mellitus in the Bio-Breeding/Worcester rat. Evidence in vitro for natural killer cell lysis of islet cells. J Clin Invest, 77, 916– 924. Michels, A.W., & Eisenbarth, G.S. (2011). Immune intervention in type 1 diabetes. Seminars in Immunology, 23, 214-219. Patterson, C.C., Dahlquist, G., Soltesz, G. et al. (2001). Is childhood-onset type I diabetes a wealth-related disease? An ecological analysis of European incidence rates. Diabetologia, 44 (Suppl 3), B9–B16. Pugliese, A. (2005). The insulin gene in type 1 diabetes. IUBMB Life, 57 (7), 463-468. Redondo, M.J., Jeffrey, J., Fain, P.R. et al. (2008). Concordance for islet autoimmunity among monozygotic twins. N Engl J Med, 359 (26), 2849–2850. Sherr, J., Sosenko, J., Skyler, J.S. et al. (2008). Prevention of type 1 diabetes: The time has come. Nat. Clin. Pract. Endocrinol. Metab., 4, 334–343. Stiller, C.R., Dupre, J., Gent, M. et al. (1987). Effects of cyclosporine in recent-onset juvenile type 1 diabetes: Impact of age and duration of disease. J Periatr, 111, 1069-1072. The DIAMOND Project Group. (2006). Incidence and trends of childhood type 1 diabetes worldwide 1990-1999. Diabet Med, 23, 857-66. Type 1 Diabetes TrialNet. (2011). Progress report on Type 1 Diabetes. Available at https://www.diabetestrialnet.org/documents/ProgressReport2011-1_2012.pdf Wagner, D. (2011). The role of T cells in type 1 diabetes. Available at http://www.intechopen.com/books/type-1-diabetes-pathogenesis-genetics-and-immunotherapy/the-role-of-t-cells-in-type-1-diabetes Wen, L., Ley, R.E., Volchkov, P.V. et al. (2008). Innate immunity and intestinal microbiota in the development of type 1 diabetes. Nature, 455, 1109–1113. Wong, M.S. et al. (2010). Gene therapy in diabetes. Self Nonself, 1 (3), 165–75. Zeigler, A., & Nepom, G. (2010). Prediction and pathogenesis in type 1 diabetes. Immunity, 32, 468-478. Suggested Reading from Inquiries Journal
Inquiries Journal provides undergraduate and graduate students around the world a platform for the wide dissemination of academic work over a range of core disciplines. Representing the work of students from hundreds of institutions around the globe, Inquiries Journal's large database of academic articles is completely free. Learn more | Blog | Submit Latest in Health Science |