Featured Article:Botulinum Toxins: Bad Bug or Miracle Medicine?
By
2010, Vol. 2 No. 11 | pg. 1/1
KEYWORDS:
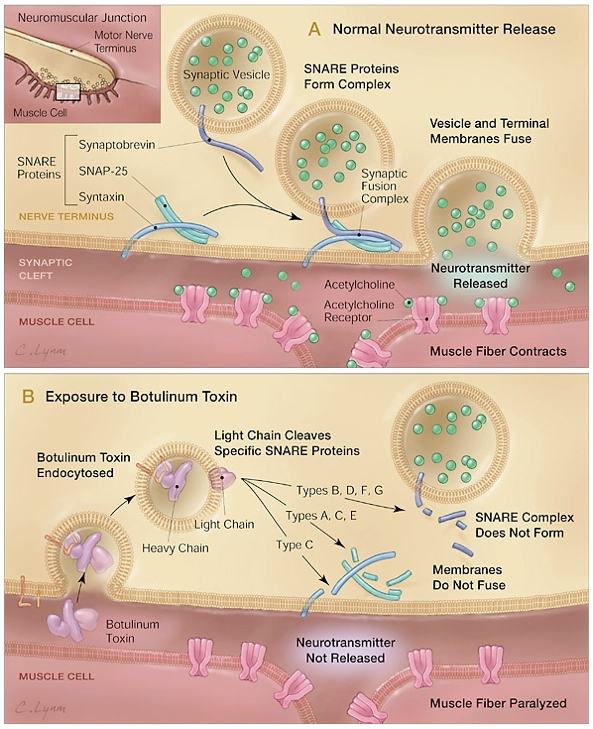
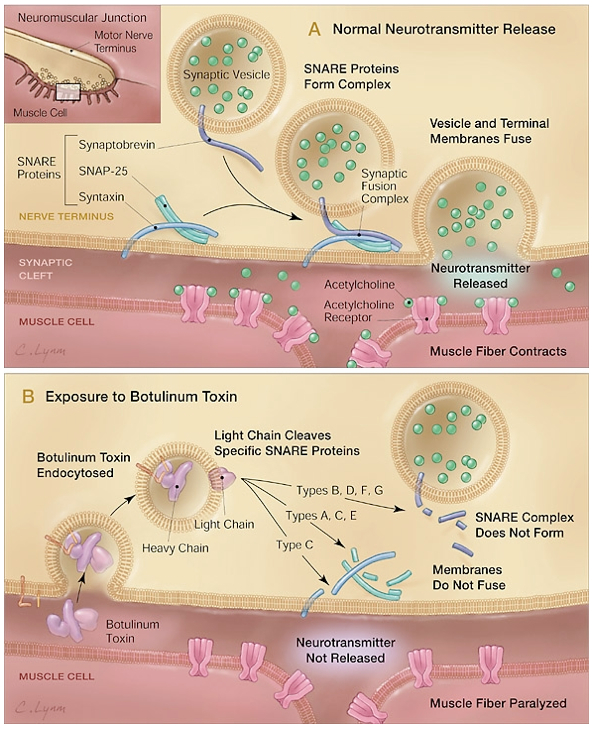
Botulinum toxin is widely known for causing a type of food poisoning known as botulism. According to the Center for Disease Control, botulism is a rare but serious paralytic illness caused by the bacterium Clostridium botulinum (Center for Disease Control, 2008). Three types of botulism exist: food borne botulism, wound botulism, and infant (intestinal) botulism. Although botulinum toxin can cause severe and often fatal illnesses, it is also used in small purified doses as a therapeutic treatment for certain medical conditions. Botulinum toxin (abbreviated as BTX or BoNT for botulinum neurotoxin) is a protein produced by the spore-forming, anaerobic, bacilli bacterium Clostridium botulinum. C. botulinum spores are found in soil and marine sediment worldwide as well as in the gastrointestinal tracts of some animals, including humans. Botulinum neurotoxins (BoNT) are some of the most toxic proteins known with a median lethal dose (LD50) of 1 ng/kg to 3 ng/kg, depending on the transmission route (Montecucco & Molgo, 2005; Koirala, n.d.). Seven serologically distinct types of BoNT have been identified and are designated A, B, C1, D, E, F, and G (Aoki, 2003). All serotypes in the BoNT family act by inhibiting the release of the neurotransmitter, acetylcholine, at the neuromuscular junction (Figure 1), thus inhibiting striated muscle contractions (Aoki, 2003; Dolly & Aoki, 2006). If botulism is not treated promptly, death can occur due to the paralysis of the respiratory muscles. Botulism was first known in the early 1800's as “sausage poison” because in Germany it was associated with eating undercooked blood sausages. In the late 1800's, the German physicist Muller, would coin the term botulism, from the Latin word botulus, which means sausage. Professor Emile Van Ermengem would be the first to isolate Clostridium botulinum in 1895. Food borne botulism has most likely occurred ever since man has tried to preserve food, however botulism was not understood and was likely attributed to other factors. Documented cases of food borne botulism became for frequent in the early 20th century due to the rise of the canned food industry. The number of botulism cases did decrease over the years as improved canning techniques were developed. Today, food borne botulism is more commonly associated with foods that are home-canned. C. botulinum spores are heat tolerant, therefore the U.S. Department of Agriculture recommends that all foods be cooked at a temperature of 100 degrees Celsius for 10 minutes.Symptoms of food borne botulism usually occur 12 to 36 hours after eating the contaminated food. Some of these symptoms may include blurred vision, dry mouth, weakness, difficulty swallowing, and paralysis. The symptoms of food borne botulism may be confused with other conditions such as Guillain-Barre syndrome, stroke, Myasynthia Gravis, tick paralysis, or intoxication. Infant botulism is rare but the most commonly occurring type of botulism. Whereas food borne botulism is contracted by ingesting the preformed BoNT, infant botulism happens when the spores of C. botulinum are ingested and the spores then produce the BoNT in the large intestine. Infant botulism usually occurs in infants between 2 weeks and 6 months of age (Baron, 1996). Infant botulism has similar symptoms as food borne botulism, but the first sign is usually constipation, which is often overlooked in infants. Infant botulism can be fatal, and is likely a cause of 4 to 15 percent of sudden infant deaths (SIDS) (Baron, 1996). Infant botulism is also known as intestinal botulism because it does occasionally occur in adults who are on antibiotic therapy or adults with a compromised GI tract. The most rare form of botulism is wound botulism. Wound botulism occurs when the spores of C. botulinum are introduced to a wound or necrotizing tissue (Passaro, Werner, McGee, MacKenzie, Vugia, 1998). The clinical signs of wound botulism are similar to the signs and symptoms of food borne and infant (intestinal) botulism. Wound botulism is most common with injecting drug users (IDU's) who inject black tar heroin (Passaro et al., 1998). Justinus Kerner, a German physician and poet, was the first to consider using botulinum toxin as a therapy for certain medical disorders. In a monograph published in 1822, Kerner described the clinical symptoms of “sausage poisoning” as well as speculating that the toxin could be used for therapeutic purposes. He hypothesized that in small doses, the botulinum toxin could be used to treat conditions that involved hyperexcitablity of the nervous system (Ergbuth, 2008). Kerner's work with this toxin would lay the foundation for future research in using BoNT as a medical drug however, further interest in using BoNT for medical purposes would not resurface until the the late 1960's (Schantz & Johnson, 1997). With the advent of World War II, the United States would begin research of biological warfare. Anthrax and BoNT were two of the top biological agents being researched at a U.S. Army facility at Camp Detrick, Maryland (now known as Fort Detrick). Botulinum toxin was known to form another type of “man-made” botulism known as inhalation botulism. Scientific researchers were attempting to to isolate and make a purified form of BoNT which would be pioneered by Carl Lamanna and James Duff (Ergbuth, 2008; Dembek, Smith, Rusnak, 2007). Working on these foundations, Edward Schantz would be the first to succeed in purifying botulinum toxin, type-A (BoNT-A) in a crystalline form (Kedlaya, 2008). While Schantz was at Fort Detrick, he was contacted in the late 1960's by Alan Scott, an eye surgeon, about the potential use of a substance that could be injected into muscles to treat strabismus. He was looking for an alternative to surgery. Strabismus, commonly known as cross-eyes, is a condition in which the eyes can not align when focusing. After Scott conducted much research with BoNT-A, first on monkeys and then humans, he concluded that BoNT-A was a successful treatment for patients with strabismus (Schantz & Johnson, 1997). Finally, in December 1989, BoNT-A (Oculinum;BOTOX) was approved by the U.S. Food and Drug Administration for the treatment of strabismus, blepharospasm, and hemifacial spasm in patients over 12 years of age. BoNT-A was the first biological toxin to be approved for the treatment of human disease. The U.S. Food and Drug Administration has approved BoNT-A (BOTOX) for the treatment of strabismus, blepharospasm, hemifacial spasm, cervical dystonia, glabellar lines (frown lines), and severe primary axillary hyperhidrosis (extreme underarm sweating). MYOBLOC (botulinum toxin type B) has also been approved by the U.S. Food and Drug Administration for treatment of adults with cervical dystonia. BoNT's have been used in various off-label uses with positive results: migraine headache, overactive bladder, benign prostatic hyperplasia, and lower back pain to name a few. Many of these off-label uses are currently in clinical trials. Although BoNT's can wreak havoc on a person with botulism, BoNT's have proven to be successful in treating medical disorders, many of which are painful or socially embarrassing. BoNT's have improved the quality of life for many, and the future use of BoNT's in medicine looks promising. Figure 1. Mechanism of Action of Botulinum Toxin (Arnon et al., 2001)
References Aoki, R.K. (2003). Evidence for Atinociceptive Activity of Botulinum Toxin Type A in Pain Management. Headache,43[suppl 1], S9-S15. Arnon S.S., Schechter R., Inglesby T.V., Henderson D.A., Bartlett J.G., Ascher M.S., et al. (2001). Botulinum Toxin as a Biological Weapon: Medical and Public Health Management [Electronic version]. JAMA, 285(8), 1059-1070. Baron S., Peake R., James D. A., Susman M., Kennedy C.A., Singleton M.D., Schuenke S. (1996). Medical Microbiology (4th ed.) Galveston, TX: The University of Texas Medical Branch at Galveston. Center for Disease Control. (2008). Botulism. Retrieved April 20, 2009, from http://www.cdc.gov/nczved/dfbmd/disease_listing/botulism_gi.html Dembek Z.F., Smith L.A., Rusnak J.M. (2007). Botulinum Toxin. In Lenhart (Ed.) & Dembek Z.F. (Vol. Ed.), Textbooks of Military Medicine: Medical Aspects of Biological Warfare. Washington, D.C.: The Borden Institute. Dolly J.O. & Aoki K.R. (2006). The structure and mode of action of different botulinum toxins [Electronic version]. European Journal of Neurology, 13[suppl. 4], 1-9. Ergbuth F.J. (2008). From poison to remedy: the chequered history of botulinum toxin [Electronic version]. Journal of Neural Transmission, 115, 559-565. Koirala J. (n.d.). Botulism: Toxicology, Clinical Presentations and Management. Retrieved April 28, 2009, from http://www.siumed.edu/medicine/infectious-diseases/current_issues/botulism.htm Kedlaya D. (2008). Botulinum Toxin: Overview. Retrieved March 19, 2009, from http://emedicine.medscape.com/article/325451-overview Montecucco C. & Molgo J. (2005). Botulinal neurotoxins: revival of an old killer. Current Opinion in Pharmacology, 5, 274-279. Retrieved May 1, 2009, from Science Direct database. Passaro D.J., Werner S.B., McGee J., MacKenzie W.R., Vugia D.J. (1998). Wound botulism associated with black tar heroin among injecting drug users [Electronic version]. JAMA, 279(11), 859-863. Schantz E.J. & Johnson E.A. (1997). Botulinum toxin: the story of its development for the treatment of human disease [Electronic version]. Perspectives in Biology and Medicine, 40(3), 317-327. Retrieved May 1, 2009 from Academic One File database. Suggested Reading from Inquiries Journal
Inquiries Journal provides undergraduate and graduate students around the world a platform for the wide dissemination of academic work over a range of core disciplines. Representing the work of students from hundreds of institutions around the globe, Inquiries Journal's large database of academic articles is completely free. Learn more | Blog | Submit Latest in Biology |