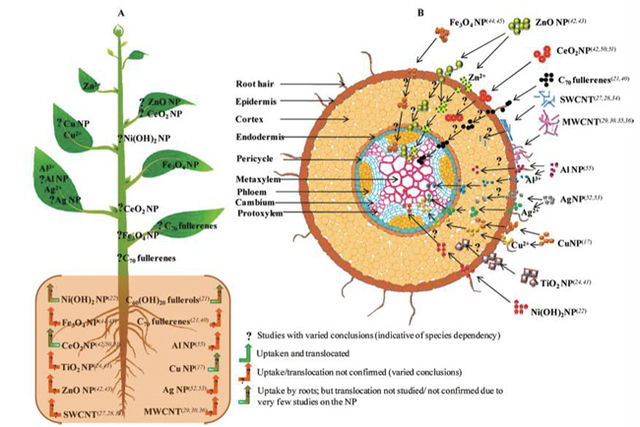
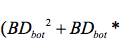From Center for Development and Strategy VOL. 2016 NO. 1Toxicological Effects of Nanomaterials on Aqueous and Terrestrial EcosystemsEvidence of Engineered NMs in Terrestrial EcosystemsNatural nanomaterials have long been recognized to exist in terrestrial ecosystems. Soil contains many materials that are less than 2 micrometers, namely "loosely called colloidal soil". (Videa et al., 2011) The components of colloidal soil contain iron oxide nanoparticles as well as humic acids and phyllosilicates. (Videa et al., 2011) However due to the increase in production of nanotechnology, various engineered nanoparticles are entering the terrestrial environment through direct modes, such as "zerovalent metal for remediation" and indirect modes such as spills and emissions. (Videa et al., 2011) Emissions are caused mainly through catalytic converters that increase the occurrence of Pt, Pd and Rh nanoparticles in the environment. (Kalavrouziotis et al., 2008) The concentrations of the nanoparticles either natural or anthropogenic are unknown in soil or terrestrial ecosystems due to the complexity of separating and identifying the nanomaterials. (Videa et al., 2011) It is assumed that the fate of the nanoparticles when they are released into such environments depends on their specific physical and chemical characteristics. (Videa et al., 2011) In addition, ionic strength, pH and soil texture also affect the impact and transport of nanomaterials, causing multiple behaviors such as "aggregation, transport, sorption, desorption, stabilization and dissolution" into the soil. (Videa et al., 2011) Uptake and Bioaccumulation of NMs in Edible PlantsRico et al., have reviewed relevant literature on the uptake and bioaccumulation of NM's in edible plants and the impacts this could have on the food chain. As previously discussed in the report, a lot of studies have gone into researching the uptake of NMs and their effects on cells, however little is known about the effects of NMs in edible plants. (Rico et al., 2011) The only carbon-based nanomaterials "shown to readily accumulate in plants" were fullerols and the C70 fullerene, whereas most of the metalbased nanomaterials such as Au, Ag, Cu and Fe were readily accumulated, "although some conflicting data exists". (Rico et al., 2011) Even though many nanomaterials are different depending on their size, shape, coating and core, data suggests that NP's can enter the plant cells by "binding to carrier proteins, through aquaporins, ion channels, or endocytosis, by creating new pores (in the case of Carbon Nanotubes (CNTs)) or by binding to organic chemicals in the environmental media". (Rico et al., 2011) Carbon based nanomaterials such as CNTs are used as novel drug delivery systems and ongoing studies are being conducted to find the mechanisms which allow CNTs to penetrate plant cells. Rico et al. (2011) hypothesize that the CNTs interact with proteins and polysaccharides on "the cell walls and elicit hypersensitive responses mimicking plant pathogens due to their small size", which ultimately leads to "cell mortality". () Metal-based (MB) nanoparticles are also studied, and very little is known on the mechanisms and uptake of MB NP's. Due to the fact that the cell wall pore sizes vary from 2-20 nm the larger nanoparticles have a harder time penetrating the cell walls, which means that only the small NP's, which were found to be more reactive, can accumulate in plants. It is also not clear if the NP's remain the same when they are inside the plant walls or if they form aggregates or colloids, which will change the function and behavior. Below is a schematic diagram of the different methods in which nanoparticles could accumulate in edible plants also showing possible mechanisms, however the data for some mechanisms is still inconclusive, as shown in the diagram. Figure 4. Diagram depicting the uptake, translocation, and biotransformation pathway of various nanoparticles in plants. Part A shows the uptake and the proposed location of the particles whereas part B shows the cross section of the absorption zone in the root, showing different nanoparticle interaction on exposure. (Rico et al., 2011) Rico et al.(2011) also concluded that the medium in which the plants are grown is important, due to an observed zero intake of NMs in plants grown in soil. Furthermore, there are no studies that show how and where plants store nanoparticles that are accumulated. Gene Expression Changes in PlantsThe effects corresponding to NM's in edible plants are found to be both positive and negative. Rico et al.(2011) give a comprehensive review of the effects different nanoparticles have on growth and germination of many plants. Van Aken et al. (2015) have also studied the gene expression changes in plants and microorganisms exposed to nanomaterials with mostly negative results. These papers concluded that the TiO2 nanoparticles on soil bacteria changed the "bacterial community structure", and showed a "reduction of nitrogen fixers and methane oxidizers" through direct toxicity to the soil. They have also concluded that many Ag nanoparticles varying in size from 45 nm to 85 nm show cell death, inhibition of cell growth, association with the cell wall, and antibacterial activity. (Van Aken, 2015) In plants, Kaveh et al., have concluded that Ag nanoparticles increase the growth of A. thaliana at low doses and decrease it at high doses, positively increasing the "pathogen resistance and plant biomass" at low doses. (Van Aken, 2015) The studies suggest that the Ag nanoparticles are affecting the plants partly through the toxicity of the Ag metal and the size of the NP. (Van Aken, 2015) (Mendes et al., 2015) Some photosynthetic pathways have also been affected by NMs in plants, specifically Ma et al., have concluded that high levels of Cerium Oxide negatively "impacted plant growth and chlorophyll production". (Ma et al., 2011) (Van Aken, 2015) Positively, it was found that when A. thaliana is exposed to TiO2 nanoparticles, a light harvesting complex gene is induced and results in increased efficiency of light absorption in the chloroplast. (Van Aken, 2015) Research has also shown that graphene oxide treated with PEG negatively impacts the development of A. thaliana seedlings due to the negative effect on genes involved in the development of the roots. Several studies, specifically Lahiani et al., have shown that multi-walled carbon nanotubes (MWCNTs) can have positive impacts on the seeds of barley, soybean and corn. Agglomerates of MWCNTs were found using Raman Spectroscopy and Transmission Electron Microscopy inside the seeds, and activated the expression of water channel genes (aquaporins) producing favorable results in growth and germination. (Lahiani et al., 2013) This study however, was created with the specific intent on increasing yield in these crops and not accounting for nanomaterials found in the environment due to unintentional release. Van Aken has also included that MWCNTs enhance the growth of tobacco over a wide range of concentrations. MWCNTs were however shown to reduce the root length of lettuce and cause cell death, plasma membrane detachment and cell shrinkage in rice. (Rico et al., 2013) Single walled carbon nanotubes (SWCNTs) have also raised concern because they inhibit growth of hair roots in maize plants. (Van Aken, 2015) Rico et al., have shown that Ag nanoparticles show reduced germination and shoot length on ryegrass, flax and barley as well as reduced transpiration and biomass on zucchini. Cu NPs were shown to have reduced seedling growth on mungbean and wheat as well as reduced root growth on zucchini. (Rico et al., 2013) CeO2 NPs show negative effects on alfalfa, tomato, lettuce, cucumber, maize and soybean through reduced shoot growth, reduced germination and reduced root growth. (Rico et al., 2013) (Doolette et al., 2015) Lastly, it is observed that nanomaterials behave differently in plants than bacteria, affecting more than one transcription factor making it difficult to understand the pathway and mechanisms involved in activation. Impacts on Humans HealthTang et al., have studied the implications that engineered nanoparticles can have on the health of infants and children. They propose that humans have a risk of coming into contact with nanomaterials through consumer products, foods, sunscreens, toys, clothes, medical applications, drug delivery and biomedical imaging. (Tang et al., 2015) Other possible mechanisms would be through bioaccumulation in the food chain through plants and crops, animals and fish as well as through direct contact with the air, water and soil. They propose that through these contact methods, nanoparticles can pose potentially threatening toxic effects for the skin when exposed to metallic nanoparticles such as iron, TiO2 and quantum dots. (Tang et al., 2015) The nanoparticles were shown to penetrate the skin barrier and cause ROS mediated skin aging, and it can also lead to "systematic exposure and development of lesions". (Tang et al., 2015) A 17-year old patient "developed hepatotoxicity and argyria-like symptoms after treatment with an Ag – containing wound dressing". (Tang et al., 2015) The respiratory system is also very susceptible to airborne nanoparticles, which can be deposited in the alveoli increasing toxic symptoms. Due to the small size of the nanoparticles, they can penetrate the "thin blood-air barrier", and move to other organs increasing the damage. In 2006, 100 German consumers had symptoms including "coughing, sleep disruption, headache and vomiting" after using a bathroom cleaning aerosol product that contained ZnO nanoparticles. (Tang et al., 2015) The illness was later found not to be linked to the nanoparticles however, a 26-year old chemist handling nickel NPs had symptoms of throat irritation, nasal congestion and flushing whereas a 38-year old male died thirteen days after inhaling nickel nanoparticles. (Tang et al., 2015). The same mechanisms that are shown in aquatic and terrestrial organisms can be expected in humans as well due to the size and shape of specific nanoparticles. Other case studies show adverse health effects to the gastrointestinal tract (GIT) and liver through exposure of engineered nanoparticles after ingesting food and pharmaceuticals. (Tang et al., 2015) Rats exposed to silver nanoparticles in the liver showed a reduced liver weight and "accumulation of granular material". (Tang et al., 2015) The brain, immune and circulation systems as well as the reproductive and developmental systems can also be affected by engineered nanoparticles. Videa et al., also discuss the impacts of engineered nanoparticles in humans and mammals showing that CuO NPs are extremely toxic to lung epithelial cells due to induced DNA and oxidative lesions. CdSe quantum dots with a coating of polyethyleneglycol (PEG) lost their coating when tested in the intestinal cells due to the lowered pH, which increased their nanotoxicity. (Videa et al., 2011) The mechanisms for exposure are the same as previously seen, where the nanoparticle size and shape allows it to enter the cells of the organs and cause toxic effects. High instances of "cardiovascular disease" were seen in workers that are handling ENPs when compared to a control group. (Tang et al., 2015) Lastly, the reproductive and developmental systems are affected "causing early miscarriages and fetal malformations" in pregnant mice after "10 days of SWCNTs injection". (Tang et al., 2015) These observed health effects on humans have large implications for future studies, showing that engineered nanomaterials should be carefully assessed before being made widely available. LimitationsThere are many limitations when trying to assess the impact that nanomaterials have on the environment. Due to the many differently engineered nanoparticles and their different sizes and properties it is very difficult to make assumptions about all nanomaterials based on the findings of one. Some nanoparticles have shown that they are extremely toxic to bacteria, yet other studies show that nanoparticles can help in the growth of edible plants. (Lahiani et al., 2013) Other limitations include individual species and their interaction with the environment as well as their ability to store or expel small nanoparticles. Furthermore future studies should focus on the long-term impacts on the environment rather than acute toxicity tests on simple species. There are also limitations in understanding how the behavior of the nanoparticles differs in the environment, and creating replicable scientific studies on the mechanisms and the bioaccumulation of nanoparticles on both aquatic and terrestrial species. (Yao et al., 2013) Very little is also discussed on the biomagnification of these nanoparticles within aquatic and terrestrial organisms and how this will affect food consumption. Moreover, fundamental studies between the structure and function relationships of nanoparticles are lacking which makes it more difficult to assess toxicity. (Rickerby et al., 2007) This study is also very wide and tries to provide a holistic view of the environmental impact of nanomaterials in many species, however in order to have a better understanding each of the aspects have to be extensively researched. ConclusionThe increase in nanotechnology poses large toxicological implications because of the release of nanomaterials into the environment affecting bacteria, edible plants, fish and mammals. The size, shape, coating and functionality of nanoparticles are crucial in understanding their specific effects on aquatic and terrestrial ecosystems. Firstly, the small size of nanoparticles increases their surface area and their ability to enter cells causing cell mortality and increased reactivity in microbes. (Suresh et al., 2013) The coating and production process have large effects on the NP characteristics, increasing toxicity and environmental impact. NMs have been shown to enter cells through cell diffusion as well as through endocytosis and adhesion. (Klein et al., 2008) This poses a threat due to the mass production of these materials making them more widely available in the atmosphere increasing the risk for plants, animals and humans to come into contact with them. Moreover, nanoparticles are shown to settle in the sediment of waterbeds as well as soil, causing small organisms living in these systems to have high nanoparticle accumulation. Lastly, human health case studies have shown that humans can be affected through inhaling, eating and coming into contact with nanoparticles affecting all the organs due to their small size and ability to enter the circulatory system. (Tang et al., 2015) However, many of the studies available are only focused on the short-term acute effects of nanoparticles in the environment, yet long-term studies are also crucial due to the fact that health impacts might not be relevant until decades later. (Tang et al., 2015) In order to study this, biomarkers can be used to examine nanoparticles in the environment and not only within the human body. (Tang et al., 2015) Finally, regulation needs to be discussed for companies that are producing nanomaterials and new technology in order to manage risk before seeing adverse health and environmental effects. Without governmental control nanomaterials are likely to accumulate in soil and water through extensive use and production of new technology, spills, runoff and emissions. ReferencesBoverhof, D. R., Bramante, C. M., Butala, J. H., Clancy, S. F., Lafranconi, M., West, J., & Gordon, S. C. (2015). Comparative assessment of nanomaterial definitions and safety evaluation considerations. Regulatory Toxicology and Pharmacology!: RTP, 73(1), 137–50. doi:10.1016/j.yrtph.2015.06.001 Burke, D. J., Pietrasiak, N., Situ, S. F., Abenojar, E. C., Porche, M., Kraj, P., … Samia, A. C. S. (2015). Iron Oxide and Titanium Dioxide Nanoparticle Effects on Plant Performance and Root Associated Microbes. International Journal of Molecular Sciences, 16(10), 23630–50. doi:10.3390/ijms161023630 Doolette, C. L., McLaughlin, M. J., Kirby, J. K., & Navarro, D. a. (2015). Bioavailability of silver and silver sulfide nanoparticles to lettuce (Lactuca sativa): Effect of agricultural amendments on plant uptake. Journal of Hazardous Materials, 300, 788–795. doi:10.1016/j.jhazmat.2015.08.012 Handy, R. D., Al-Bairuty, G., Al-Jubory, a, Ramsden, C. S., Boyle, D., Shaw, B. J., & Henry, T. B. (2011). Effects of manufactured nanomaterials on fishes: a target organ and body systems physiology approach. Journal of Fish Biology, 79(4), 821–53. doi:10.1111/j.1095-8649.2011.03080.x Kalavrouziotis, I. K., & Koukoulakis, P. H. (2008). The Environmental Impact of the Platinum Group Elements (Pt, Pd, Rh) Emitted by the Automobile Catalyst Converters. Water, Air, and Soil Pollution, 196(1-4), 393–402. doi:10.1007/s11270-008-9786-9 Lowry, G. V., Hotze, E. M., Bernhardt, E. S., Dionysiou, D. D., Pedersen, J. a., Wiesner, M. R., & Xing, B. (2010). Environmental Occurrences, Behavior, Fate, and Ecological Effects of Nanomaterials: An Introduction to the Special Series. Journal of Environment Quality, 39(6), 1867. doi:10.2134/jeq2010.0297 MacCormack, T. J., & Goss, G. G. (2008). Identifying and Predicting Biological Risks Associated With Manufactured Nanoparticles in Aquatic Ecosystems. Journal of Industrial Ecology, 12(3), 286–296. doi:10.1111/j.1530-9290.2008.00041.x Mehndiratta, P., Jain, A., Srivastava, S., & Gupta, N. (2013). Environmental Pollution and Nanotechnology. Environment and Pollution, 2(2), 49–59. doi:10.5539/ep.v2n2p49 Mendes, L. A., Maria, V. L., Scott-Fordsmand, J. J., & Amorim, M. J. B. (2015). Ag Nanoparticles (Ag NM300K) in the Terrestrial Environment: Effects at Population and Cellular Level in Folsomia candida (Collembola). International Journal of Environmental Research and Public Health, 12(10), 12530–42. doi:10.3390/ijerph121012530 Peijnenburg, W. (2011). Guest editorial. Environmental fate and effects of nanoparticles. Environment International, 37(6), 1043. doi:10.1016/j.envint.2011.04.001 Peralta-Videa, J. R., Zhao, L., Lopez-Moreno, M. L., de la Rosa, G., Hong, J., & Gardea-Torresdey, J. L. (2011). Nanomaterials and the environment: a review for the biennium 2008-2010. Journal of Hazardous Materials, 186(1), 1–15. doi:10.1016/j.jhazmat.2010.11.020 Rickerby, D. G., & Morrison, M. (2007). Nanotechnology and the environment: A European perspective. Science and Technology of Advanced Materials, 8(1-2), 19–24. doi:10.1016/j.stam.2006.10.002 Griffitt, Arber, D. A. S. B. (2008). Effects of particle composition and species on toxicity of metallic nanomaterials in aquatic organisms. 27(9), 1972–1978. Smita, S., Gupta, S. K., Bartonova, A., Dusinska, M., Gutleb, A. C., & Rahman, Q. (2012). Nanoparticles in the environment: assessment using the causal diagram approach. Environmental Health!: A Global Access Science Source, 11 Suppl 1(Suppl 1), S13. doi:10.1186/1476-069X-11-S1-S13 Suresh, A. K., Pelletier, D. a, & Doktycz, M. J. (2013). Relating nanomaterial properties and microbial toxicity. Nanoscale, 5(2), 463–74. doi:10.1039/c2nr32447d Tang, S., Wang, M., Germ, K. E., Du, H.-M., Sun, W.-J., Gao, W.-M., & Mayer, G. D. (2015). Health implications of engineered nanoparticles in infants and children. World Journal of Pediatrics!: WJP, 11(3), 197–206. doi:10.1007/s12519-015-0028-0 Yao, D., Chen, Z., Zhao, K., Yang, Q., & Zhang, W. (2013). Limitation and Challenge Faced to the Researches on Environmental Risk of Nanotechnology. Procedia Environmental Sciences, 18, 149– 156. doi:10.1016/j.proenv.2013.04.020 Van Aken, B. (2015). Gene expression changes in plants and microorganisms exposed to nanomaterials. Current Opinion in Biotechnology, 33, 206–19. doi:10.1016/j.copbio.2015.03.005 Hou, W.-C., Westerhoff, P., & Posner, J. D. (2013). Biological accumulation of engineered nanomaterials: a review of current knowledge. Environ. Sci.: Processes Impacts, 15(1), 103–122. doi:10.1039/C2EM30686G Holden, P. A., Nisbet, R. M., Lenihan, H. S., Miller, R. J., Cherr, G. N., Schimel, J. P., & Gardeatorresdey, J. L. (2013). Ecological Nanotoxicology!: Integrating Nanomaterial Hazard Considerations Across, 46(3), 813–822. Rico, C. M., Majumdar, S., Duarte-Gardea, M., Peralta-Videa, J. R., & Gardea-Torresdey, J. L. (2011). Interaction of nanoparticles with edible plants and their possible implications in the food chain. Journal of Agricultural and Food Chemistry, 59(8), 3485–98. doi:10.1021/jf104517j Lahiani, M. H., Dervishi, E., Chen, J., Nima, Z., Gaume, A., Biris, A. S., & Khodakovskaya, M. V. (2013). Impact of carbon nanotube exposure to seeds of valuable crops. ACS Applied Materials & Interfaces, 5(16), 7965–73. doi:10.1021/am402052x Linkov, I., Bates, M. E., Canis, L. J., Seager, T. P., & Keisler, J. M. (2011). A decision-directed approach for prioritizing research into the impact of nanomaterials on the environment and human health. Nature Nanotechnology, 6(12), 784–787. doi:10.1038/nnano.2011.163 Suggested Reading from Inquiries Journal
Inquiries Journal provides undergraduate and graduate students around the world a platform for the wide dissemination of academic work over a range of core disciplines. Representing the work of students from hundreds of institutions around the globe, Inquiries Journal's large database of academic articles is completely free. Learn more | Blog | Submit Latest in Environmental Studies |