Carbon Capture and Storage as a Method to Mitigate Climate Change
By
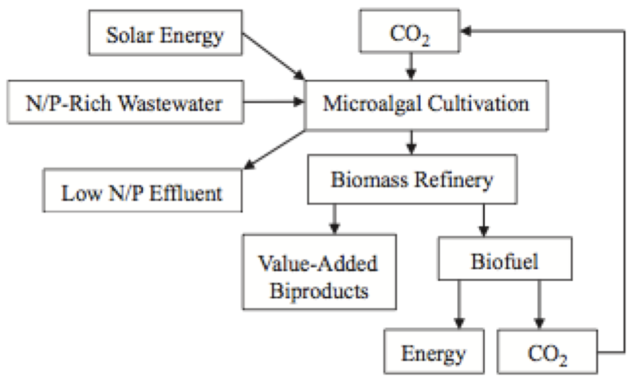
2017, Vol. 9 No. 03 | pg. 2/2 | « Biological Carbon Sequestration MethodsWhile the various methods of physical capture and storage of CO2 are very valuable and can be used in existing plants and industries worldwide, it is important to have both physical and biological approaches to combat climate change. Biological carbon mitigation involves biological media, specifically autotrophic organisms and plants, which utilize carbon fixation as a food source by converting carbon dioxide to glucose. (Farrelly et al., 2013) This is the well-known process of photosynthesis where plants use energy from light to form glucose in their chlorophyll containing tissues. (Farrelly et al., 2013) Glucose is then stored as starch and cellulose in plants creating organic carbon and sequestering it from the environment. (Farrelly et al., 2013) Moreover, natural terrestrial and aquatic sequestration methods can also be enhanced in order to fully create a low carbon future in which anthropogenic sources are balanced within the carbon cycle. Terrestrial Carbon SequestrationDuring photosynthesis the carbon from CO2 is turned into carbohydrates for plant growth and is later released during decomposition. However, some of this carbon is naturally sequestered in the soil or in wetlands providing a method to store anthropogenic sources of CO2. (Litynski et al., 2006) The world’s forest ecosystems cover about 4.1 billion hectares globally and are a very large reserve for terrestrial carbon sequestration. (Lal et al., 2005) The major anthropogenic factors that may affect the soil organic carbon within forests are “forest management activities, deforestation, afforestation of agricultural soils and subsequent management of forest plantations.” (Lal et al., 2005) In order to increase the carbon sequestration ability of forests and terrestrial ecosystems it is very crucial that management techniques are considered to increase the capture of carbon by plants and effectively store it. Soil carbon sequestration technologies also exist, such as using biochar in addition to the soil in order to increase the carbon capture capacity. (Smith, 2016) AfforestationAfforestation/reforestation and a specific choice of species are crucial in enhancing the soil organic carbon, meaning that it will enhance the sequestration ability of the land. (Lal et al., 2005) A study was done assessing the different land use systems and their organic carbon stock, concluding that the “the total C stock was 128 Mg/ha under pasture, 173 Mg/ha under 29-year-old forest and 232 Mg/ha under 69-year-old stand.” (Lal et al., 2005) Moreover, they discovered that “the total C stock was 117 Mg/ha under 27-year-old pin oak stand and 227 Mg/ha under 69-year-old oak beech stand,” concluding that age is very important in forest management practices and increase of carbon sequestration. (Lal et al., 2005) Afforestation is a technique that can be used and has been used in redeveloping old mine sites, brownfields and other areas that did not have any capacity to store carbon dioxide. However, even though forests have a large role to play in CO2 storage, the impacts of climate change will alter the ecosystem productivity, allocation of the biomass, and microbial populations, which can affect ultimately, affect carbon storage. (Lal, 2005)Ocean Carbon SequestrationThe ocean holds 50 times more inorganic carbon than the atmosphere and acts as a natural carbon sink, sequestering a net 2 GtC per year. (Farrelly et al., 2013) This occurs through one of two processes, photosynthesis or a solubility pump. Sequestration through photosynthesis occurs when the photosynthetic organisms die and sink “by vertical flux to the deep ocean” and the fixated carbon within them remains in the sediment at the bottom of the ocean. (Farrelly et al., 2013) The solubility pump acts as a process of sequestration by exploiting the solubility of carbon dioxide in water forming carbonates. These carbonates are then transported through convection currents and ocean currents to the deep ocean remaining for long periods of time. (Farrelly et al., 2013) The limiting factor in biological carbon sequestration through the oceans is the nutrient availability, initiating various studies to see if ocean enrichment can be possible in order to mitigate climate change. These initiatives have led to the creation of pilot projects using ammonia as a nutrient in order to enhance the phytoplankton growth in the upper ocean to sequester carbon efficiently and biologically. (Otaegui, 2016) Moreover, ocean carbon sequestration can also occur on a geological timeline of hundreds and thousands of years, where the carbon is stored in the form of Magnesium and Calcium salts using the reactions below. (Farrelly et al., 2013) CO2 + CaSiO3 <--> CaCO3 +SiO2 (1) CO2 + MgSi <--> MgCO3 +SiO2 (2) These are the inorganic reactions that are driven by organic processes in algae and ocean bacteria, which can also be used as a process to sequester carbon. Ocean carbon sequestration does have a large downfall due to the process of ocean acidification, which occurs with the increase of carbonic acid in the oceans due to a high CO2 saturation. This also will lead to a maximum rate of sequestration ability by oceans at net 5 GtG/year by 2100. (Farrelly et al., 2013) Furthermore, if one chooses to implement ocean nourishment by iron or nitrogen, it has the ability to change the structure of plankton having a long-term impact on ocean ecosystems. If large quantities of biomass sink to the bottom of the ocean, this could trigger methane gas production effectively counteracting any positive carbon fixation effect. (Farrelly et al., 2013) Biological Carbon MitigationMicroalgaeBiological carbon mitigation (BCM) occurs through natural processes and using organisms and photosynthesis to sequester carbon. Microalgae have been studied for decades as a feedstock for renewable energy in order to mitigate climate change. (Bhola et al., 2014) They are able to use concentrated carbon dioxide, which comes from power plants as well as various other sources, to produce different types of biomass representing a very powerful greenhouse gas mitigation strategy. (Bhola et al., 2014) The efficiency of the microalgae is related to the CO2 concentration in the growth atmosphere, with a higher concentration resulting in higher productivity. (Singh et al., 2013) Yet the supply of carbon to the microalgal mass culture systems is a limitation that must be solved for this technology. (Singh et al., 2013) Choosing the right strains of microalgae for CO2 mitigation is very crucial in terms of efficiency and cost competitiveness with previous studies showing that the tolerance and concentration differ significantly between strains. (Singh et al., 2013) What has been observed is that the value of CO2 concentration required for growth is a lot less than the microalgal strain tolerance for CO2. Singh et al., give a few examples of this observation by stating that the Euglena gracilis is 42% CO2 tolerant, yet the best growth was observed at 5% CO2 concentration. Growth is important because through the production of biomass, there is a possibility to use it as a renewable energy source both mitigating carbon and creating an efficient energy system. (Farrelly et al., 2013) A conceptual map of a microalgal system to create renewable energy and also sequester carbon is shown in Figure 5. Figure 5. Conceptual map of a microalgal system combinging biofuel production, CO2 mitigation and N/P removal from wastewater. (Farrelly et al., 2013). Figure 5 shows the various uses that microalgal cultivation can have on a large scale basis. Through the input of solar energy, nutrient rich wastewater and CO2 the algae can be cultivated through a biomass refinery, creating biofuel, bi products, energy and the ability to capture the CO2 and reuse it for another process. The culturing on an industrial scale can be costly, it has large potential to produce fuel from direct combustion, gasification, pyrolysis, anaerobic digestion and other chemical processes. (Farrelly et al., 2014) Microalgal photosynthesis can also result in creating calcium carbonate, which can act as a long-term storage of carbon dioxide. (Farrelly et al., 2013) Lastly, not only is this method of carbon sequestration a lot faster it has also proved to be more efficient than using higher plants on various levels. (Bhola et al., 2014) The resilience to adverse conditions, CO2 fixation capacity, production and harversting time, scale-up process, and acid deposition were studied comparing microalgae to terrestrial plants concluding that algae is more resilient, has a higher fixation capacity, can be produced all year round and has no acid deposition. (Bhola et al., 2014) CyanobacteriaCyanobacteria can also be used as a biological carbon mitigation method, due to the fact that they are photoautotrophic fast growing unicellular organisms that use CO2 as their food source. (Farrelly et al., 2013) They require anaerobic conditions, and grow at a temperature of 50 to 75 0C. The reaction typically needs light and an absence of N2, in order to have good growth and produce hydrogen as a byproduct. (Farrelly et al., 21013) Similarly to the microalgae, cyanobacteria can be a form of sustainable energy because they can biofixate the CO2 in a photobioreactor and reuse the CO2 to form lipids, which in turn can be made into biodiesel. This effectively mitigates carbon dioxide and creates energy. (Farrelly et al., 2013) A few strains were found to be extremely effective with a theoretical uptake of one tonne of carbon dioxide per hour if the bioreactor was scaled to 4000 m3. (Farrelly et al., 2013) One of the major issues with this type of method of sequestration is the cost, and the novelty of using cyanobacteria over other proven methods. LimitationsMany of the technological limitations lie in the cost and economic analysis of the process. Usually the technology is very young, and still in the development phase rendering it useless to mitigate carbon dioxide in large-scale industrial facilities. (Leung et al., 2013) There is also a lack of a market mechanism/incentive “that is sufficiently large and long term” to reward companies to use carbon capture and storage technologies. (Leung et al., 2013) Furthermore, the storage practices that are mature are not as reliable as deep saline aquifer storage or ocean storage however those are still in the implementation and research phase in their development. Forest management and terrestrial ecosystems can be improved to increase the sequestration capacity; however there will always be a maximum amount of C that can be stored. This also has a cost associated with it, which can be a potential barrier or limitation for many people. (Singh et al., 2013) Lastly, Microalgal limitations occur during the harvesting process, which contributes 20-30% to the cost of microalgal “biomass production.” (Singh et al., 2013) The microalgal culture will also have a higher energy demand in industrial scale processes. (Singh et al., 2013) Cyanobacteria on the other hand, are very limited due to its novelty and not enough research. The high cost associated with microalgae production and cyanobacterial growth is a limiting factor in trying to scale the systems, as well as the microalgae culture has a low absorption of CO2 and there is a lack of relevant expertise as “the algal industry is very young.” (Singh et al., 2013) ConclusionTo conclude, carbon sequestration methods are a viable option in order to help mitigate climate change and reduce atmospheric carbon emissions. Many ecosystems are at risk of being altered due to climate change, such as various land changes, increased natural disasters, ocean acidification and others, which will impact not only the population but also various natural environmental processes. The first method discussed as a way to capture carbon dioxide was physical sequestration, specifically involving new and existing carbon capture and storage technologies. Carbon capture, separation and storage technologies have been used in order to attempt the mitigation of the GHG emissions from anthropogenic activities, such as large point source emissions from power plants and industrial processes. (Singh et al., 2013) Carbon capture technologies includes chemical capture during the pre combustion stage, post combustion as well as during oxyfuel combustion, followed by separation then storage of the CO2. The biological methods discussed mostly focused on microalgae and cyanobacteria as a suitable example for carbon utilization and storage. (Kumar et al., 2011) Moreover, terrestrial sequestration through afforestation and land use management is also an important method in order to increase the C stock in soil. Oceans also play a large role in mitigation, holding most of the worlds carbon, however many limitations exist in carbon capture and increase of C saturation in the ocean waters due to acidification through the increase in carbonic acid. Lastly, microalgae can be used as a method to mitigate climate change and show a lot of promise due to their faster growth and high photosynthetic activity. (Singh et al., 2013) ReferencesAbanades, J. C., Arias, B., Lyngfelt, a., Mattisson, T., Wiley, D. E., Li, H., … Brandani, S. (2015). Emerging CO2 capture systems. International Journal of Greenhouse Gas Control, 40, 126–166. doi:10.1016/j.ijggc.2015.04.018 Bhola, V., Swalaha, F., Ranjith Kumar, R., Singh, M., & Bux, F. (2014). Overview of the potential of microalgae for CO2 sequestration. International Journal of Environmental Science and Technology, 11(7), 2103–2118. doi:10.1007/s13762-013-0487-6 Brunetti, a., Scura, F., Barbieri, G., & Drioli, E. (2010). Membrane technologies for CO2 separation. Journal of Membrane Science, 359(1-2), 115–125. doi:10.1016/j.memsci.2009.11.040 Farrelly, D. J., Everard, C. D., Fagan, C. C., & McDonnell, K. P. (2013). Carbon sequestration and the role of biological carbon mitigation: A review. Renewable and Sustainable Energy Reviews, 21, 712–727. doi:10.1016/j.rser.2012.12.038 Gebreslassie, B. H., Waymire, R., & You, F. (2013). Sustainable design and synthesis of algae-based biorefinery for simultaneous hydrocarbon biofuel production and carbon sequestration. AIChE Journal, 59(5), 1599–1621. doi:10.1002/aic.14075 Kirkby, C. a., Richardson, A. E., Wade, L. J., Batten, G. D., Blanchard, C., & Kirkegaard, J. a. (2013). Carbon-nutrient stoichiometry to increase soil carbon sequestration. Soil Biology and Biochemistry, 60, 77–86. doi:10.1016/j.soilbio.2013.01.011 Kumar, K., Dasgupta, C. N., Nayak, B., Lindblad, P., & Das, D. (2011). Development of suitable photobioreactors for CO2 sequestration addressing global warming using green algae and cyanobacteria. Bioresource Technology, 102(8), 4945–53. doi:10.1016/j.biortech.2011.01.054 Lal, R. (2004). Soil carbon sequestration impacts on global climate change and food security. Science (New York, N.Y.), 304(5677), 1623–7. doi:10.1126/science.1097396 Lal, R. (2005). Forest soils and carbon sequestration. Forest Ecology and Management, 220(1-3), 242–258. doi:10.1016/j.foreco.2005.08.015 Lemieux, J.-M. (2011). Review: The potential impact of underground geological storage of carbon dioxide in deep saline aquifers on shallow groundwater resources. Hydrogeology Journal, 19(4), 757–778. doi:10.1007/s10040-011-0715-4 Leung, D. Y. C., Caramanna, G., & Maroto-Valer, M. M. (2014). An overview of current status of carbon dioxide capture and storage technologies. Renewable and Sustainable Energy Reviews, 39, 426–443. doi:10.1016/j.rser.2014.07.093 Mundschau, M. V., Xie, X., Evenson, C. R., & Sammells, a. F. (2006). Dense inorganic membranes for production of hydrogen from methane and coal with carbon dioxide sequestration. Catalysis Today, 118(1-2), 12–23. doi:10.1016/j.cattod.2006.01.042 Muradov, N., & Veziroglu, T. (2008). “Green” path from fossil-based to hydrogen economy: An overview of carbon-neutral technologies. International Journal of Hydrogen Energy, 33(23), 6804–6839. doi:10.1016/j.ijhydene.2008.08.054 Powlson, D. S., Whitmore, a. P., & Goulding, K. W. T. (2011). Soil carbon sequestration to mitigate climate change: a critical re-examination to identify the true and the false. European Journal of Soil Science, 62(1), 42–55. doi:10.1111/j.1365-2389.2010.01342.x Singh, U. B., & Ahluwalia, a. S. (2012). Microalgae: a promising tool for carbon sequestration. Mitigation and Adaptation Strategies for Global Change, 18(1), 73–95. doi:10.1007/s11027-012-9393-3 Smith, P. (2016). Soil carbon sequestration and biochar as negative emission technologies. Global Change Biology, 1315–1324. doi:10.1111/gcb.13178 Standish, R. J., & Hulvey, K. B. (2014). Co-benefits of planting species mixes in carbon projects. Ecological Management & Restoration, 15(1), 26–29. doi:10.1111/emr.12084 Stanger, R., Wall, T., Spörl, R., Paneru, M., Grathwohl, S., Weidmann, M., … Santos, S. (2015). Oxyfuel combustion for CO2 capture in power plants. International Journal of Greenhouse Gas Control, 40, 55–125. doi:10.1016/j.ijggc.2015.06.010 Thomson, A. M., César Izaurralde, R., Smith, S. J., & Clarke, L. E. (2008). Integrated estimates of global terrestrial carbon sequestration. Global Environmental Change, 18(1), 192–203. doi:10.1016/j.gloenvcha.2007.10.002 Van Oosterzee, P. (2012). The integration of biodiversity and climate change: A contextual assessment of the carbon farming initiative. Ecological Management & Restoration, 13(3), 238–244. doi:10.1111/emr.12001 Suggested Reading from Inquiries Journal
Inquiries Journal provides undergraduate and graduate students around the world a platform for the wide dissemination of academic work over a range of core disciplines. Representing the work of students from hundreds of institutions around the globe, Inquiries Journal's large database of academic articles is completely free. Learn more | Blog | Submit Latest in Environmental Studies |